Endothelial differentiation of bone marrow mesenchyme stem cells applicable to hypoxia and increased migration through Akt and NFκB signals
- PMID: 28173835
- PMCID: PMC5296962
- DOI: 10.1186/s13287-017-0470-0
Endothelial differentiation of bone marrow mesenchyme stem cells applicable to hypoxia and increased migration through Akt and NFκB signals
Abstract
Background: Bone marrow mesenchymal stem cells (MSCs) and endothelial progenitor cells (EPCs) are used to repair hypoxic or ischemic tissue. However, the underlining mechanism of resistance in the hypoxic microenvironment and the efficacy of migration to the injured tissue are still unknown. The current study aims to understand the hypoxia resistance and migration ability of MSCs during differentiation toward endothelial lineages by biochemical and mechanical stimuli.
Method: MSCs were harvested from the bone marrow of 6-8-week-old Sprague-Dawley rats. The endothelial growth medium (EGM) was added to MSCs for 3 days to initiate endothelial differentiation. Laminar shear stress was used as the fluid mechanical stimulation.
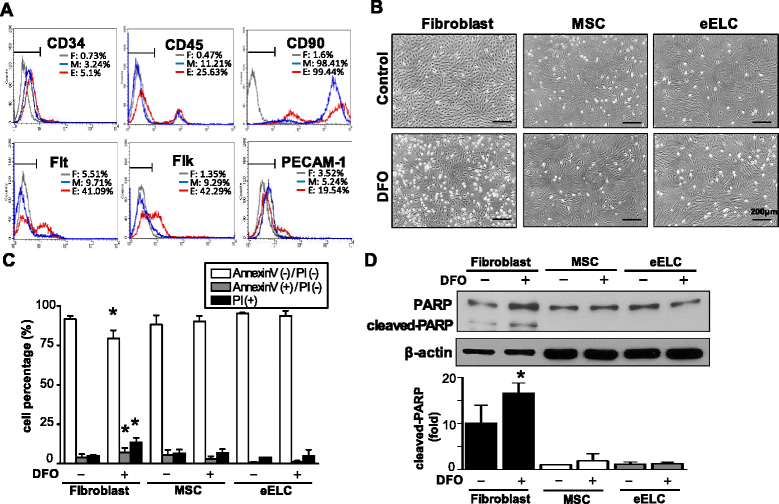
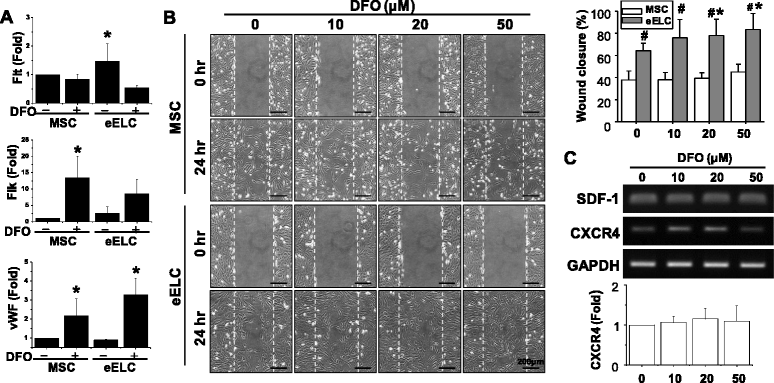
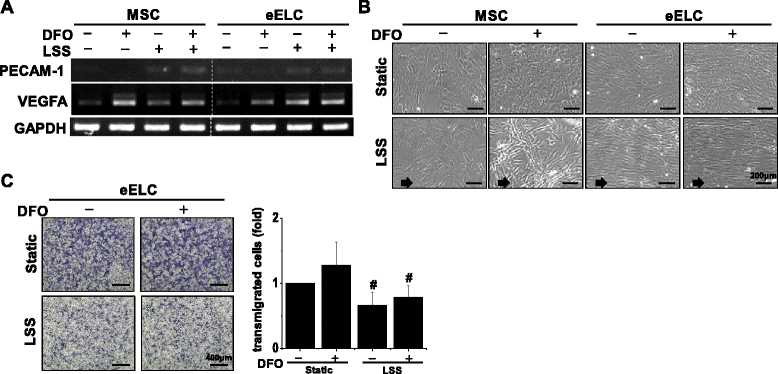
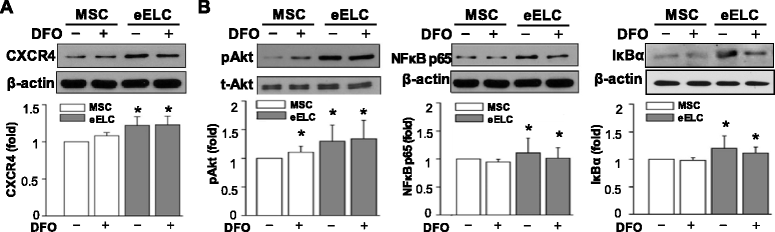
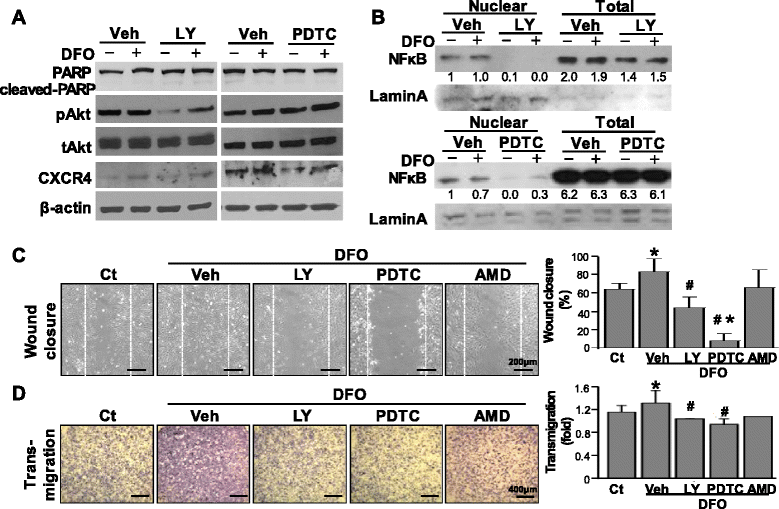
Results: Application of EGM facilitated the early endothelial lineage cells (eELCs) to express EPC markers. When treating the hypoxic mimetic desferrioxamine, both MSCs and eELCs showed resistance to hypoxia as compared with the occurrence of apoptosis in rat fibroblasts. The eELCs under hypoxia increased the wound closure and C-X-C chemokine receptor type 4 (CXCR4) gene expression. Although the shear stress promoted eELC maturation and aligned cells parallel to the flow direction, their migration ability was not superior to that of eELCs either under normoxia or hypoxia. The eELCs showed higher protein expressions of CXCR4, phosphorylated Akt (pAkt), and endogenous NFκB and IκBα than MSCs under both normoxia and hypoxia conditions. The potential migratory signals were discovered by inhibiting either Akt or NFκB using specific inhibitors and revealed decreases of wound closure and transmigration ability in eELCs.
Conclusion: The Akt and NFκB pathways are important to regulate the early endothelial differentiation and its migratory ability under a hypoxic microenvironment.
Keywords: Akt; Endothelial differentiation; Hypoxia; Migration; Nuclear factor-κB; Stem cell.
Figures
Similar articles
-
α-Tocopherol, especially α-tocopherol phosphate, exerts antiapoptotic and angiogenic effects on rat bone marrow-derived endothelial progenitor cells under high-glucose and hypoxia conditions.J Vasc Surg. 2018 Apr;67(4):1263-1273.e1. doi: 10.1016/j.jvs.2017.02.051. Epub 2017 May 29. J Vasc Surg. 2018. PMID: 28571880
-
The angiogenic related functions of bone marrow mesenchymal stem cells are promoted by CBDL rat serum via the Akt/Nrf2 pathway.Exp Cell Res. 2016 May 15;344(1):86-94. doi: 10.1016/j.yexcr.2016.04.013. Epub 2016 Apr 19. Exp Cell Res. 2016. PMID: 27105936
-
Peptide hormone ELABELA promotes rat bone marrow-derived mesenchymal stem cell proliferation and migration by manipulating the cell cycle through the PI3K/AKT pathway under the hypoxia and ischemia microenvironment.Stem Cell Res Ther. 2022 Jan 28;13(1):32. doi: 10.1186/s13287-021-02691-1. Stem Cell Res Ther. 2022. PMID: 35090551 Free PMC article.
-
Effects of shear stress on endothelial progenitor cells.J Biomed Nanotechnol. 2014 Oct;10(10):2586-97. doi: 10.1166/jbn.2014.2014. J Biomed Nanotechnol. 2014. PMID: 25992410 Review.
-
A possible new focus for stroke treatment - migrating stem cells.Expert Opin Biol Ther. 2015 Jul;15(7):949-58. doi: 10.1517/14712598.2015.1043264. Epub 2015 May 5. Expert Opin Biol Ther. 2015. PMID: 25943632 Free PMC article. Review.
Cited by
-
The atypical RhoGTPase RhoE/Rnd3 is a key molecule to acquire a neuroprotective phenotype in microglia.J Neuroinflammation. 2018 Dec 15;15(1):343. doi: 10.1186/s12974-018-1386-z. J Neuroinflammation. 2018. PMID: 30553270 Free PMC article.
-
Effects of shear stress on differentiation of stem cells into endothelial cells.World J Stem Cells. 2021 Jul 26;13(7):894-913. doi: 10.4252/wjsc.v13.i7.894. World J Stem Cells. 2021. PMID: 34367483 Free PMC article. Review.
-
Mesenchymal Stem Cell Migration and Tissue Repair.Cells. 2019 Jul 28;8(8):784. doi: 10.3390/cells8080784. Cells. 2019. PMID: 31357692 Free PMC article. Review.
-
Female Age Affects the Mesenchymal Stem Cell Characteristics of Aspirated Follicular Cells in the In Vitro Fertilization Programme.Stem Cell Rev Rep. 2019 Aug;15(4):543-557. doi: 10.1007/s12015-019-09889-0. Stem Cell Rev Rep. 2019. PMID: 31055736 Clinical Trial.
-
Mesenchymal stem cells: a promising way in therapies of graft-versus-host disease.Cancer Cell Int. 2020 Apr 7;20:114. doi: 10.1186/s12935-020-01193-z. eCollection 2020. Cancer Cell Int. 2020. PMID: 32280306 Free PMC article. Review.
References
-
- Saito A, Maier CM, Narasimhan P, Nishi T, Song YS, Yu F, Liu J, Lee YS, Nito C, Kamada H, Dodd RL, Hsieh LB, Hassid B, Kim EE, Gonzalez M, Chan PH. Oxidative stress and neuronal death/survival signaling in cerebral ischemia. Mol Neurobiol. 2005;31(1–3):105–116. doi: 10.1385/MN:31:1-3:105. - DOI - PubMed
-
- Matsushita H, Morishita R, Nata T, Aoki M, Nakagami H, Taniyama Y, Yamamoto K, Higaki J, Yasufumi K, Ogihara T. Hypoxia-induced endothelial apoptosis through nuclear factor-kappaB (NF-kappaB)-mediated bcl-2 suppression: in vivo evidence of the importance of NF-kappaB in endothelial cell regulation. Circ Res. 2000;86(9):974–981. doi: 10.1161/01.RES.86.9.974. - DOI - PubMed
-
- Aoki M, Nata T, Morishita R, Matsushita H, Nakagami H, Yamamoto K, Yamazaki K, Nakabayashi M, Ogihara T, Kaneda Y. Endothelial apoptosis induced by oxidative stress through activation of NF-kappaB: antiapoptotic effect of antioxidant agents on endothelial cells. Hypertension. 2001;38(1):48–55. doi: 10.1161/01.HYP.38.1.48. - DOI - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources

