Imaging the adult zebrafish cone mosaic using optical coherence tomography
- PMID: 28177275
- PMCID: PMC5659228
- DOI: 10.1017/S0952523816000092
Imaging the adult zebrafish cone mosaic using optical coherence tomography
Erratum in
-
Imaging the adult zebrafish cone mosaic using optical coherence tomography-CORRIGENDUM.Vis Neurosci. 2017 Jan;34:E005. doi: 10.1017/S0952523817000025. Vis Neurosci. 2017. PMID: 28965523 Free PMC article. No abstract available.
Abstract
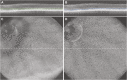
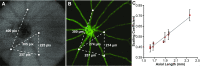
Zebrafish (Danio rerio) provide many advantages as a model organism for studying ocular disease and development, and there is great interest in the ability to non-invasively assess their photoreceptor mosaic. Despite recent applications of scanning light ophthalmoscopy, fundus photography, and gonioscopy to in vivo imaging of the adult zebrafish eye, current techniques either lack accurate scaling information (limiting quantitative analyses) or require euthanizing the fish (precluding longitudinal analyses). Here we describe improved methods for imaging the adult zebrafish retina using spectral domain optical coherence tomography (OCT). Transgenic fli1:eGFP zebrafish were imaged using the Bioptigen Envisu R2200 broadband source OCT with a 12-mm telecentric probe to measure axial length and a mouse retina probe to acquire retinal volume scans subtending 1.2 × 1.2 mm nominally. En face summed volume projections were generated from the volume scans using custom software that allows the user to create contours tailored to specific retinal layer(s) of interest. Following imaging, the eyes were dissected for ex vivo fluorescence microscopy, and measurements of blood vessel branch points were compared to those made from the en face OCT images to determine the OCT lateral scale as a function of axial length. Using this scaling model, we imaged the photoreceptor layer of five wild-type zebrafish and quantified the density and packing geometry of the UV cone submosaic. Our in vivo cone density measurements agreed with measurements from previously published histology values. The method presented here allows accurate, quantitative assessment of cone structure in vivo and will be useful for longitudinal studies of the zebrafish cone mosaics.
Keywords: Cone mosaic; Optical coherence tomography; Retina; Zebrafish.
Figures


Similar articles
-
Repeatability and Reproducibility of In Vivo Cone Density Measurements in the Adult Zebrafish Retina.Adv Exp Med Biol. 2018;1074:151-156. doi: 10.1007/978-3-319-75402-4_19. Adv Exp Med Biol. 2018. PMID: 29721939 Free PMC article.
-
Spectral Domain Optical Coherence Tomography: An In Vivo Imaging Protocol for Assessing Retinal Morphology in Adult Zebrafish.Zebrafish. 2017 Apr;14(2):118-125. doi: 10.1089/zeb.2016.1376. Epub 2017 Jan 4. Zebrafish. 2017. PMID: 28051361
-
Ontogeny of cone photoreceptor mosaics in zebrafish.J Comp Neurol. 2010 Oct 15;518(20):4182-95. doi: 10.1002/cne.22447. J Comp Neurol. 2010. PMID: 20878782 Free PMC article.
-
A moving wave patterns the cone photoreceptor mosaic array in the zebrafish retina.Int J Dev Biol. 2004;48(8-9):935-45. doi: 10.1387/ijdb.041873pr. Int J Dev Biol. 2004. PMID: 15558484 Review.
-
Genetic approaches to retinal research in zebrafish.J Neurogenet. 2017 Sep;31(3):70-87. doi: 10.1080/01677063.2017.1343316. Epub 2017 Jul 5. J Neurogenet. 2017. PMID: 28678567 Review.
Cited by
-
Novel segmentation algorithm for high-throughput analysis of spectral domain-optical coherence tomography imaging of teleost retinas.Mol Vis. 2022 Dec 31;28:492-499. eCollection 2022. Mol Vis. 2022. PMID: 37089699 Free PMC article.
-
Multicontrast investigation of in vivo wildtype zebrafish in three development stages using polarization-sensitive optical coherence tomography.J Biomed Opt. 2022 Jan;27(1):016001. doi: 10.1117/1.JBO.27.1.016001. J Biomed Opt. 2022. PMID: 35064657 Free PMC article.
-
Neurodegeneration, Neuroprotection and Regeneration in the Zebrafish Retina.Cells. 2021 Mar 12;10(3):633. doi: 10.3390/cells10030633. Cells. 2021. PMID: 33809186 Free PMC article. Review.
-
Imaging the adult zebrafish cone mosaic using optical coherence tomography-CORRIGENDUM.Vis Neurosci. 2017 Jan;34:E005. doi: 10.1017/S0952523817000025. Vis Neurosci. 2017. PMID: 28965523 Free PMC article. No abstract available.
-
The Effect of Retinal Melanin on Optical Coherence Tomography Images.Transl Vis Sci Technol. 2017 Apr 3;6(2):8. doi: 10.1167/tvst.6.2.8. eCollection 2017 Apr. Transl Vis Sci Technol. 2017. PMID: 28392975 Free PMC article.
References
-
- Allison W.T., Haimberger T.J., Hawryshyn C.W. & Temple S.E. (2004). Visual pigment composition in zebrafish: Evidence for a rhodopsin-porphyropsin interchange system. Visual Neuroscience 21, 945–952. - PubMed
-
- Branchek T. & Bremiller R. (1984). The development of photoreceptors in the zebrafish, Brachydanio rerio. I. Structure. Journal of Comparative Neurology 224, 107–115. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials

