Anaplastic thyroid carcinoma and foscarnet use in a multitarget treatment documented by 18F-FDG PET/CT: A case report
- PMID: 28178124
- PMCID: PMC5312981
- DOI: 10.1097/MD.0000000000005621
Anaplastic thyroid carcinoma and foscarnet use in a multitarget treatment documented by 18F-FDG PET/CT: A case report
Abstract
Rationale: The case reported the rapid remission of disease recurrence achieved adding foscarnet, a DNA polymerase inhibitor that interacts with fibroblast growth factor 2, to low molecular weight heparin and sunitinib for the first time in a patient with an anaplastic thyroid cancer (ATC).
Patient concerns: A 65-year-old woman with a multinodular goiter referred for a rapid enlargement of a nodule. Histological examination revealed an ATC with a little area of papillary thyroid cancer (PTC). The patient was resistant to selective single-target treatment.
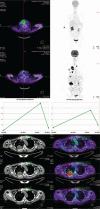
Diagnoses: Immunophenotyping and gene analyses found a significant increase in FGF2 and FGFR1 expression in the primary ATC area (FGF2 = 38.2 ± 6.2% in ATC vs 34.6 ± 6.0% in the differentiated area of PTC, P < 0.05; FGFR1: 41.7 ± 6.0% in ATC vs 34.4 ± 4.2% in PTC, P < 0.001) and in metastatic neck lymph nodes (P < 0.001 vs normal control tissues). Unlike conventional imaging, F-FDG PET/CT with PERCIST 1.0 criteria promptly and quantitatively detected disease recurrence and remission before and after multitarget therapy, combining anatomic, metabolic, and functional data.
Interventions: Foscarnet was administered given the positivity for FGF2, FGFR1 and FGFR4 in ATC. Low molecular wight heparin and Sunitinib were coadministere to limiti metastatic progression and on neck tumor masse, respectively.
Outcomes: The rationale for the clinical response to this innovative multitarget association with foscarnet is based on the histological and genetic finding that fibroblast growth factors and their receptor super-family are up-regulated in the primary anaplastic thyroid tumor and in the metastatic lymph node of our patient.
Lessons: We propose that fibroblast growth factors and their receptor super-family play a key role as potential therapeutic targets in anaplastic thyroid cancer and the positive relevance of this suggestion for patient care, especially for an individualized management.
Conflict of interest statement
The authors have no conflicts of interest to disclose.
Figures


Similar articles
-
Non‑invasive monitoring of paclitaxel and lenvatinib efficacy against anaplastic thyroid cancer in orthotopic SCID mouse models using small‑animal FDG‑PET/CT.Oncol Rep. 2020 Oct;44(4):1709-1716. doi: 10.3892/or.2020.7720. Epub 2020 Aug 7. Oncol Rep. 2020. PMID: 32945510
-
FGF-Receptors and PD-L1 in Anaplastic and Poorly Differentiated Thyroid Cancer: Evaluation of the Preclinical Rationale.Front Endocrinol (Lausanne). 2021 Aug 12;12:712107. doi: 10.3389/fendo.2021.712107. eCollection 2021. Front Endocrinol (Lausanne). 2021. PMID: 34475850 Free PMC article.
-
Anaplastic thyroid carcinoma with rapid thyrotoxicosis - a case report and the literature review.Endokrynol Pol. 2018;69(1):28-31. doi: 10.5603/EP.a2018.0010. Epub 2018 Jan 10. Endokrynol Pol. 2018. PMID: 29319130 Review.
-
Complete response to weekly carboplatin-docetaxel chemotherapy in a 91-year-old woman with anaplastic thyroid cancer.Am J Otolaryngol. 2015 Mar-Apr;36(2):268-72. doi: 10.1016/j.amjoto.2014.03.006. Epub 2014 Mar 27. Am J Otolaryngol. 2015. PMID: 25681954 Review.
-
PROGNOSTIC VALUE OF LYMPH NODE UPTAKE ON PRETREATMENT F-18 FDG PET/CT IN PATIENTS WITH N1B PAPILLARY THYROID CARCINOMA.Endocr Pract. 2019 Aug;25(8):787-793. doi: 10.4158/EP-2018-0607. Epub 2019 Apr 23. Endocr Pract. 2019. PMID: 31013158
Cited by
-
Actions of Thyroid Hormones on Thyroid Cancers.Front Endocrinol (Lausanne). 2021 Jun 21;12:691736. doi: 10.3389/fendo.2021.691736. eCollection 2021. Front Endocrinol (Lausanne). 2021. PMID: 34234745 Free PMC article. Review.
-
Ouabain Effects on Human Anaplastic Thyroid Carcinoma 8505C Cells.Cancers (Basel). 2022 Dec 14;14(24):6168. doi: 10.3390/cancers14246168. Cancers (Basel). 2022. PMID: 36551653 Free PMC article.
-
Predictors of Response and Survival to Multikinase Inhibitors in Radioiodine Resistant Differentiated Thyroid Cancer.J Pers Med. 2021 Jul 18;11(7):674. doi: 10.3390/jpm11070674. J Pers Med. 2021. PMID: 34357141 Free PMC article. Review.
-
Prognostic Role of Pre-Treatment [18F]FDG PET/CT in Patients with Anaplastic Thyroid Cancer.Cancers (Basel). 2021 Aug 23;13(16):4228. doi: 10.3390/cancers13164228. Cancers (Basel). 2021. PMID: 34439382 Free PMC article.
References
-
- Sosa JA, Elisei R, Jarzab B, et al. Randomized safety and efficacy study of fosbretabulin with paclitaxel/carboplatin against anaplastic thyroid carcinoma. Thyroid 2014;24:232–40. - PubMed
-
- Sosa JA, Balkissoon J, Lu SP, et al. Thyroidectomy followed by fosbretabulin (CA4P) combination regimen appears to suggest improvement in patient survival in anaplastic thyroid cancer. Surgery 2012;152:1078–87. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials
Miscellaneous

