Translation and Clinical Development of Bispecific T-cell Engaging Antibodies for Cancer Treatment
- PMID: 28182247
- PMCID: PMC5763312
- DOI: 10.1002/cpt.651
Translation and Clinical Development of Bispecific T-cell Engaging Antibodies for Cancer Treatment
Abstract
Bispecific T-cell Engagers (BiTE®) antibody constructs enable a polyclonal T-cell response to cell-surface tumor-associated antigens, bypassing the narrow specificities of T-cell receptors and the need for antigen presentation through the major histocompatibility complex pathways. Blinatumomab, a CD19xCD3 BiTE® antibody construct, received accelerated approval for the treatment of relapsed/refractory Philadelphia chromosome negative acute lymphoblastic leukemia. Herein we review the pharmacology, safety, and efficacy observed in studies of blinatumomab and other BiTE® antibody constructs. Quantitative systems pharmacology is envisioned as a means to optimize dosing decisions for trials in which BiTE® antibody constructs are administered as monotherapy or in combination with other immunotherapies.
© 2017 The Authors. Clinical Pharmacology & Therapeutics published by Wiley Periodicals, Inc. on behalf of American Society for Clinical Pharmacology and Therapeutics.
Figures
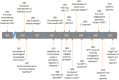
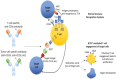

References
-
- Coley, W.B. The treatment of malignant tumors by repeated inoculations of erysipelas. With a report of ten original cases. 1893. Clin. Orthopaed. Relat. Res. 262, 3–11 (1991). - PubMed
-
- Ehrlich, P. Ueber den jetzigen Stand der Karzinomforschung. Ned. Tijdschr. Geneeskd 5, 273–290 (1909).
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources

