Localization of Short-Chain Polyphosphate Enhances its Ability to Clot Flowing Blood Plasma
- PMID: 28186112
- PMCID: PMC5301195
- DOI: 10.1038/srep42119
Localization of Short-Chain Polyphosphate Enhances its Ability to Clot Flowing Blood Plasma
Abstract
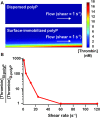
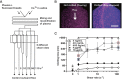
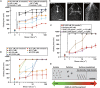
Short-chain polyphosphate (polyP) is released from platelets upon platelet activation, but it is not clear if it contributes to thrombosis. PolyP has increased propensity to clot blood with increased polymer length and when localized onto particles, but it is unknown whether spatial localization of short-chain polyP can accelerate clotting of flowing blood. Here, numerical simulations predicted the effect of localization of polyP on clotting under flow, and this was tested in vitro using microfluidics. Synthetic polyP was more effective at triggering clotting of flowing blood plasma when localized on a surface than when solubilized in solution or when localized as nanoparticles, accelerating clotting at 10-200 fold lower concentrations, particularly at low to sub-physiological shear rates typical of where thrombosis occurs in large veins or valves. Thus, sub-micromolar concentrations of short-chain polyP can accelerate clotting of flowing blood plasma under flow at low to sub-physiological shear rates. However, a physiological mechanism for the localization of polyP to platelet or vascular surfaces remains unknown.
Conflict of interest statement
S.A.S., D.K., G.D.S., and J.H.M. are co-inventors on pending patent applications covering potential medical uses of SNP-polyP. A.J.D. and Y.L. are co-inventors on a pending patent application related to the therapeutic usage and delivery of NP-polyP. D.K., G.D.S., and J.H.M. declare competing financial interests in this work. The remaining authors declare no competing financial interests.
Figures

References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

