Monocytic angiotensin-converting enzyme 2 relates to atherosclerosis in patients with chronic kidney disease
- PMID: 28186543
- PMCID: PMC7108029
- DOI: 10.1093/ndt/gfw206
Monocytic angiotensin-converting enzyme 2 relates to atherosclerosis in patients with chronic kidney disease
Abstract
Background: Increased levels of monocytic angiotensin-converting enzyme (ACE) found in haemodialysis (HD) patients may directly participate in the pathogenesis of atherosclerosis. We demonstrated recently that uremia triggers the development of highly pro-atherogenic monocytes via an angiotensin II (AngII)–dependent mechanism. Opposing actions of the AngII-degrading ACE2 remain largely unknown. We examined the status of both ACEs and related receptors in circulating leukocytes of HD, not-dialyzed CKD and healthy individuals. Furthermore, we tested the possible impact of monocytic ACEs on atherogenesis and behaviour of the cells under conditions mimicking chronic renal failure.
Methods: Expression of ACE, ACE2, AT1R, AT2R and MASR was investigated on circulating leukocytes from 71 HD (62 ± 14 years), 24 CKD stage 3–5 (74 ± 10 years) patients and 37 healthy control subjects (53 ± 6 years) and isolated healthy monocytes treated with normal and uremic serum. Analyses of ACE, ACE2, ICAM-1, VCAM-1, MCSF and endothelial adhesion were tested on ACE-overexpressing THP-1 monocytes treated with captopril or losartan. ACE2-overexpressing monocytes were subjected to transmigration and adhesion assays and investigated for MCP-1, ICAM-1, VCAM-1, MCSF, AT1R and AT2R expression.
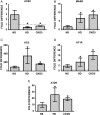
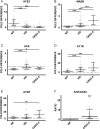
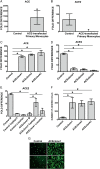
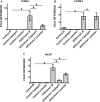
Results: The ACE mRNA level was significantly increased in HD and CKD stage 3–5 leukocytes. Correspondingly, ACE2 was downregulated and AngII as well as MAS receptor expression was upregulated in these cells. Healthy monocytes preconditioned with uremic serum reflected the same expressional regulation of ACE/ACE2, MAS and AngII receptors as those observed in HD and CKD stage 3–5 leukocytes. Overexpression of monocytic ACE dramatically decreased levels of ACE2 and induced a pro-atherogenic phenotype, partly reversed by AngII-modifying treatments, leading to an increase in ACE2. Overexpression of ACE2 in monocytes led to reduced endothelial adhesion, transmigration and downregulation of adhesion-related molecules.
Conclusions: HD and not-dialyzed CKD stage 3–5 patients show enhanced ACE and decreased ACE2 expression on monocytes. This constellation renders the cells endothelial adhesive and likely supports the development of atherosclerosis.
Keywords: ACE2; atherosclerosis; CKD; monocytes; THP-1; uremia.
Figures


References
-
- Cheung AK, Sarnak MJ, Yan G et al. Atherosclerotic cardiovascular disease risks in chronic hemodialysis patients. Kidney Int 2000; 58: 353–362 - PubMed
-
- Vanholder R, Massy Z, Argiles A et al. Chronic kidney disease as cause of cardiovascular morbidity and mortality. Nephrol Dial Transplant 2005; 20: 1048–1056 - PubMed
-
- Guerin AP, Blacher J, Pannier B et al. Impact of aortic stiffness attenuation on survival of patients in end-stage renal failure. Circulation 2001; 103: 987–992 - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials
Miscellaneous

