The Potential of MicroRNAs as Novel Biomarkers for Transplant Rejection
- PMID: 28191475
- PMCID: PMC5278203
- DOI: 10.1155/2017/4072364
The Potential of MicroRNAs as Novel Biomarkers for Transplant Rejection
Abstract
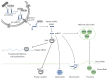
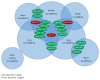
The control of gene expression by microRNAs (miRNAs, miR) influences many cellular functions, including cellular differentiation, cell proliferation, cell development, and functional regulation of the immune system. Recently, miRNAs have been detected in serum, plasma, and urine and circulating miR profiles have been associated with a variety of diseases. Rejection is one of the major causes of allograft failure and preventing and treating acute rejection are the central task for clinicians working with transplant patients. Invasive biopsies used in monitoring rejection are burdensome and risky to transplant patients. Novel and easily accessible biomarkers of acute rejection could make it possible to detect rejection earlier and make more fine-tuned calibration of immunosuppressive or new target treatment possible. In this review, we discuss whether circulating miRNA can serve as an early noninvasive diagnostic biomarker and an expression fingerprint of allograft rejection and transplant failure. Understanding the regulatory interplay of relevant miRNAs and the rejecting allograft will result in a better understanding of the molecular pathophysiology of alloimmune injury.
Conflict of interest statement
The authors declare that they have no competing interests.
Figures
References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources

