Age-related reduction of dermal fibroblast size upregulates multiple matrix metalloproteinases as observed in aged human skin in vivo
- PMID: 28196296
- PMCID: PMC5555832
- DOI: 10.1111/bjd.15379
Age-related reduction of dermal fibroblast size upregulates multiple matrix metalloproteinases as observed in aged human skin in vivo
Abstract
Background: Fragmentation of collagen fibrils, the major structure protein in skin, is a hallmark of dermal ageing. Matrix metalloproteinases (MMPs) are largely responsible for the fragmentation of collagen fibrils.
Objectives: To quantify gene expression of all 23 known mammalian MMPs in sun-protected young and aged human skin in vivo and to investigate the potential mechanism underlying age-related alteration of multiple MMPs.
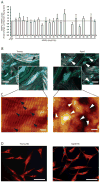
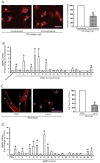
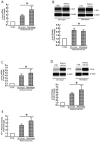
Methods: MMP mRNA expression levels and MMP activity in sun-protected young and aged human skin in vivo were determined by real-time reverse transcription polymerase chain reaction (RT-PCR) and in situ zymography, respectively. The relative contributions to elevated MMPs in epidermis and dermis were quantified by laser capture microdissection coupled real-time RT-PCR. Dermal fibroblast morphology and collagen fibril fragmentation in human skin in vivo were assessed by second-harmonic generation microscopy and atomic force microscopy, respectively. In vitro cell morphology was assessed by CellTracker® fluorescent dye (Molecular Probes, Eugene, OR, U.S.A.) and phalloidin staining. Protein levels were determined by ProteinSimple capillary electrophoresis immunoassay (ProteinSimple, Santa Clare, CA, U.S.A.).
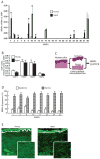
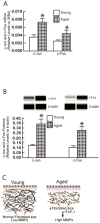
Results: Multiple MMPs are elevated in aged human skin dermis. Increased MMP activity and collagen fibril fragmentation were observed in aged skin dermis. As dermal fibroblasts are the major MMP-producing cells in the dermis, reduction of dermal fibroblast size, which is observed in aged human skin, contributes to the elevation of age-related multiple MMPs. Reduction of fibroblast size upregulates c-Jun/c-Fos and activates AP-1.
Conclusions: Combined actions of the wide variety of MMPs that are constitutively elevated in aged dermis may be involved in the progressive degradation of dermal collagen fibrils. Age-related elevations of multiple MMPs are likely to be a result of the reduction of fibroblast size via activation of AP-1.
© 2017 British Association of Dermatologists.
Figures

Comment in
-
Extracellular matrix is an influential force in ageing.Br J Dermatol. 2017 Nov;177(5):1160-1161. doi: 10.1111/bjd.15980. Br J Dermatol. 2017. PMID: 29192971 No abstract available.
Similar articles
-
Cell-size-dependent upregulation of HGF expression in dermal fibroblasts: Impact on human skin connective tissue aging.J Dermatol Sci. 2017 Dec;88(3):289-297. doi: 10.1016/j.jdermsci.2017.08.003. Epub 2017 Aug 4. J Dermatol Sci. 2017. PMID: 28826691
-
Age-associated reduction of cellular spreading/mechanical force up-regulates matrix metalloproteinase-1 expression and collagen fibril fragmentation via c-Jun/AP-1 in human dermal fibroblasts.Aging Cell. 2014 Dec;13(6):1028-37. doi: 10.1111/acel.12265. Epub 2014 Sep 9. Aging Cell. 2014. PMID: 25201474 Free PMC article.
-
Ultraviolet irradiation induces CYR61/CCN1, a mediator of collagen homeostasis, through activation of transcription factor AP-1 in human skin fibroblasts.J Invest Dermatol. 2010 Jun;130(6):1697-706. doi: 10.1038/jid.2010.29. Epub 2010 Feb 18. J Invest Dermatol. 2010. PMID: 20164845
-
Matrix-degrading metalloproteinases in photoaging.J Investig Dermatol Symp Proc. 2009 Aug;14(1):20-4. doi: 10.1038/jidsymp.2009.8. J Investig Dermatol Symp Proc. 2009. PMID: 19675548 Free PMC article. Review.
-
Molecular mechanisms of photoaging and its prevention by retinoic acid: ultraviolet irradiation induces MAP kinase signal transduction cascades that induce Ap-1-regulated matrix metalloproteinases that degrade human skin in vivo.J Investig Dermatol Symp Proc. 1998 Aug;3(1):61-8. J Investig Dermatol Symp Proc. 1998. PMID: 9732061 Review.
Cited by
-
The Role of Probiotics in Skin Photoaging and Related Mechanisms: A Review.Clin Cosmet Investig Dermatol. 2022 Nov 16;15:2455-2464. doi: 10.2147/CCID.S388954. eCollection 2022. Clin Cosmet Investig Dermatol. 2022. PMID: 36420112 Free PMC article. Review.
-
The Skin Microbiome: Current Landscape and Future Opportunities.Int J Mol Sci. 2023 Feb 16;24(4):3950. doi: 10.3390/ijms24043950. Int J Mol Sci. 2023. PMID: 36835363 Free PMC article. Review.
-
Molecular Mechanisms of Changes in Homeostasis of the Dermal Extracellular Matrix: Both Involutional and Mediated by Ultraviolet Radiation.Int J Mol Sci. 2022 Jun 15;23(12):6655. doi: 10.3390/ijms23126655. Int J Mol Sci. 2022. PMID: 35743097 Free PMC article. Review.
-
YAP/TAZ regulates TGF-β/Smad3 signaling by induction of Smad7 via AP-1 in human skin dermal fibroblasts.Cell Commun Signal. 2018 Apr 25;16(1):18. doi: 10.1186/s12964-018-0232-3. Cell Commun Signal. 2018. PMID: 29695252 Free PMC article.
-
Biomarkers of aging.Sci China Life Sci. 2023 May;66(5):893-1066. doi: 10.1007/s11427-023-2305-0. Epub 2023 Apr 11. Sci China Life Sci. 2023. PMID: 37076725 Free PMC article. Review.
References
-
- Di Lullo GA, Sweeney SM, Korkko J, et al. Mapping the ligand-binding sites and disease-associated mutations on the most abundant protein in the human, type I collagen. J Biol Chem. 2002;277:4223–31. - PubMed
-
- Uitto J. Connective tissue biochemistry of the aging dermis. Age-related alterations in collagen and elastin. Dermatol Clin. 1986;4:433–46. - PubMed
-
- Ma W, Wlaschek M, Tantcheva-Poor I, et al. Chronological ageing and photoageing of the fibroblasts and the dermal connective tissue. Clin Exp Dermatol. 2001;26:592–9. - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials
Miscellaneous

