Novel Variant in the ANK2 Membrane-Binding Domain Is Associated With Ankyrin-B Syndrome and Structural Heart Disease in a First Nations Population With a High Rate of Long QT Syndrome
- PMID: 28196901
- PMCID: PMC5312931
- DOI: 10.1161/CIRCGENETICS.116.001537
Novel Variant in the ANK2 Membrane-Binding Domain Is Associated With Ankyrin-B Syndrome and Structural Heart Disease in a First Nations Population With a High Rate of Long QT Syndrome
Abstract
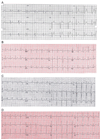
Background: Long QT syndrome confers susceptibility to ventricular arrhythmia, predisposing to syncope, seizures, and sudden death. While rare globally, long QT syndrome is ≈15× more common in First Nations of Northern British Columbia largely because of a known mutation in KCNQ1. However, 2 large multigenerational families were affected, but negative for the known mutation.
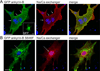
Methods and results: Long QT syndrome panel testing was carried out in the index case of each family, and clinical information was collected. Cascade genotyping was performed. Biochemical and myocyte-based assays were performed to evaluate the identified gene variant for loss-of-function activity. Index cases in these 2 families harbored a novel ANK2 c.1937C>T variant (p.S646F). An additional 16 carriers were identified, including 2 with structural heart disease: one with cardiomyopathy resulting in sudden death and the other with congenital heart disease. For all carriers of this variant, the average QTc was 475 ms (±40). Although ankyrin-B p.S646F is appropriately folded and expressed in bacteria, the mutant polypeptide displays reduced expression in cultured H9c2 cells and aberrant localization in primary cardiomyocytes. Furthermore, myocytes expressing ankyrin-B p.S646F lack normal membrane targeting of the ankyrin-binding partner, the Na/Ca exchanger. Thus, ankyrin-B p.S646F is a loss-of-function variant.
Conclusions: We identify the first disease-causing ANK2 variant localized to the membrane-binding domain resulting in reduced ankyrin-B expression and abnormal localization. Further study is warranted on the potential association of this variant with structural heart disease given the role of ANK2 in targeting and stabilization of key structural and signaling molecules in cardiac cells.
Keywords: Wolff–Parkinson–White syndrome; arrhythmia; genetic variation; heart rate; long QT syndrome.
© 2017 American Heart Association, Inc.
Figures





Comment in
-
Ankyrin-B Defects: Serendipity and Inquisitiveness are the Mothers of Invention.Circ Cardiovasc Genet. 2017 Jan;10(1):e001698. doi: 10.1161/CIRCGENETICS.117.001698. Circ Cardiovasc Genet. 2017. PMID: 28196903 No abstract available.
Similar articles
-
Defining the cellular phenotype of "ankyrin-B syndrome" variants: human ANK2 variants associated with clinical phenotypes display a spectrum of activities in cardiomyocytes.Circulation. 2007 Jan 30;115(4):432-41. doi: 10.1161/CIRCULATIONAHA.106.656512. Epub 2007 Jan 22. Circulation. 2007. PMID: 17242276
-
A cardiac arrhythmia syndrome caused by loss of ankyrin-B function.Proc Natl Acad Sci U S A. 2004 Jun 15;101(24):9137-42. doi: 10.1073/pnas.0402546101. Epub 2004 Jun 3. Proc Natl Acad Sci U S A. 2004. PMID: 15178757 Free PMC article.
-
Common human ANK2 variant confers in vivo arrhythmia phenotypes.Heart Rhythm. 2016 Sep;13(9):1932-40. doi: 10.1016/j.hrthm.2016.06.012. Epub 2016 Jun 11. Heart Rhythm. 2016. PMID: 27298202
-
Ankyrin-based cardiac arrhythmias: a new class of channelopathies due to loss of cellular targeting.Curr Opin Cardiol. 2005 May;20(3):189-93. doi: 10.1097/01.hco.0000160372.95116.3e. Curr Opin Cardiol. 2005. PMID: 15861006 Review.
-
Cardiac ankyrins in health and disease.J Mol Cell Cardiol. 2009 Aug;47(2):203-9. doi: 10.1016/j.yjmcc.2009.04.010. Epub 2009 Apr 24. J Mol Cell Cardiol. 2009. PMID: 19394342 Free PMC article. Review.
Cited by
-
Reappraisal of ANK2 Variants in Cardiovascular Diseases: Uncovering Mechanisms and Future Directions.Rev Cardiovasc Med. 2025 Jan 15;26(1):26013. doi: 10.31083/RCM26013. eCollection 2025 Jan. Rev Cardiovasc Med. 2025. PMID: 39867173 Free PMC article. Review.
-
Ion channel traffic jams: the significance of trafficking deficiency in long QT syndrome.Cell Discov. 2025 Jan 10;11(1):3. doi: 10.1038/s41421-024-00738-0. Cell Discov. 2025. PMID: 39788950 Free PMC article. Review.
-
A mild phenotype associated with KCNQ1 p.V205M mediated long QT syndrome in First Nations children of Northern British Columbia: effect of additional variants and considerations for management.Front Pediatr. 2024 May 31;12:1394105. doi: 10.3389/fped.2024.1394105. eCollection 2024. Front Pediatr. 2024. PMID: 38884101 Free PMC article.
-
Indigenous Genomic Databases: Pragmatic Considerations and Cultural Contexts.Front Public Health. 2020 Apr 24;8:111. doi: 10.3389/fpubh.2020.00111. eCollection 2020. Front Public Health. 2020. PMID: 32391301 Free PMC article.
-
Study of pathogenic genes in a pedigree with familial dilated cardiomyopathy.World J Clin Cases. 2023 Apr 16;11(11):2412-2422. doi: 10.12998/wjcc.v11.i11.2412. World J Clin Cases. 2023. PMID: 37123301 Free PMC article.
References
MeSH terms
Substances
Supplementary concepts
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

