Long-Term Treatment of Native LDL Induces Senescence of Cultured Human Endothelial Cells
- PMID: 28197300
- PMCID: PMC5288541
- DOI: 10.1155/2017/6487825
Long-Term Treatment of Native LDL Induces Senescence of Cultured Human Endothelial Cells
Erratum in
-
Erratum to "Long-Term Treatment of Native LDL Induces Senescence of Cultured Human Endothelial Cells".Oxid Med Cell Longev. 2017;2017:4576985. doi: 10.1155/2017/4576985. Epub 2017 Sep 20. Oxid Med Cell Longev. 2017. PMID: 29098060 Free PMC article.
Abstract
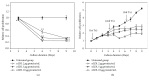
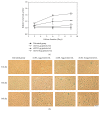
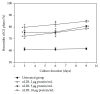
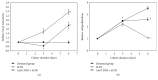
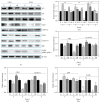
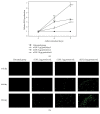
The study aimed to evaluate whether the treatment of primary cultured human endothelial cells with native low-density lipoprotein (nLDL) could induce their senescence and to uncover some of the putative mechanisms involved. For this purpose, human umbilical vein endothelial cells (HUVECs) were subcultured and/or continuously cultured with nLDL (0, 2, 5, and 10 μg protein/mL), for up to 9 days. The results indicated that nLDL inhibited the proliferation of HUVECs by arresting the cell cycle at G1 phase. The G1-arrested cells showed increase in cytosolic senescence-associated-β-galactosidase (SA-β-Gal) activity, a biomarker of cellular senescence. The causative factor of the cellular senescence was nLDL itself and not oxidized LDL (oxLDL), since blocking LDL receptor (LDLR) with the anti-LDLR antibody opposed the nLDL-induced increase of SA-β-Gal activity and decrease of cellular proliferation. In addition, nLDL-induced cellular senescence by inhibiting the phosphorylation of pRb (G1 arrest) via p53 as well as p16 signal transduction pathways. G1 phase arrest of the senescent cells was not overcome by nLDL removal from the culture medium. Moreover, the nLDL-treated cells produced reactive oxygen species (ROS) dose- and time-dependently. These results suggested, for the first time, that long-term treatment of nLDL could induce the premature senescence of endothelial cells.
Conflict of interest statement
The authors declare that there is no conflict of interests regarding the publication of this paper.
Figures
References
-
- Celermajer D. S., Sorensen K. E., Spiegelhalter D. J., Georgakopoulos D., Robinson J., Deanfield J. E. Aging is associated with endothelial dysfunction in healthy men years before the age-related decline in women. Journal of the American College of Cardiology. 1994;24(2):471–476. doi: 10.1016/0735-1097(94)90305-0. - DOI - PubMed
-
- Eggen D. A., Solberg L. A. Variation of atherosclerosis with age. Laboratory Investigation. 1968;18(5):571–579. - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous

