Redistribution of soluble N-ethylmaleimide-sensitive-factor attachment protein receptors in mouse sperm membranes prior to the acrosome reaction
- PMID: 28203732
- PMCID: PMC8483236
- DOI: 10.1095/biolreprod.116.143735
Redistribution of soluble N-ethylmaleimide-sensitive-factor attachment protein receptors in mouse sperm membranes prior to the acrosome reaction
Abstract
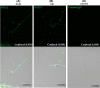
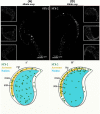
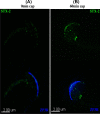
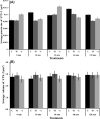
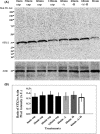
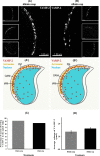
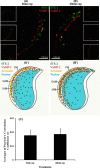
Formation of complexes between soluble N-ethylmaleimide-sensitive-factor attachment protein receptor (SNARE) proteins on opposing membranes is the minimal requirement for intracellular membrane fusion. The SNARE, syntaxin 2, is found on the sperm plasma membrane and a second SNARE, vesicle associated membrane protein 2 (VAMP2, also known as synaptobrevin 2, SYB2), is on the apposing outer acrosomal membrane. During the acrosome reaction, the outer acrosomal membrane fuses at hundreds of points with the plasma membrane. We hypothesized that syntaxin 2 and VAMP2 redistribute within their respective membranes prior to the acrosome reaction to form trans-SNARE complexes and promote membrane fusion. Immunofluorescence and superresolution structured illumination microscopy were used to localize syntaxin 2 and VAMP2 in mouse sperm during capacitation. Initially, syntaxin 2 was found in puncta throughout the acrosomal region. At 60 and 120 min of capacitation, syntaxin 2 was localized in puncta primarily in the apical ridge. Although deletion of bicarbonate during incubation had no effect, syntaxin 2 puncta were relocated in the restricted region in less than 20% of sperm incubated without albumin. In contrast, VAMP2 was already found in puncta within the apical ridge prior to capacitation. The puncta containing syntaxin 2 and VAMP2 did not precisely co-localize at 0 or 60 min of capacitation time. In summary, syntaxin 2 shifted its location to the apical ridge on the plasma membrane during capacitation in an albumin-dependent manner but VAMP2 was already localized to the apical ridge. Puncta containing VAMP2 did not co-localize with those containing syntaxin 2 during capacitation; therefore, formation of trans-SNARE complexes containing these SNAREs does not occur until after capacitation, immediately prior to acrosomal exocytosis.
Keywords: SNAREs; sperm; acrosome reaction; exocytosis; capacitation; syntaxin 2 (STX2); vesicle associated membrane protein 2 (VAMP2); membrane fusion; plasma membrane.
Figures


Similar articles
-
Human sperm capacitation is necessary for SNARE assembly in neurotoxin-resistant complexes.Andrology. 2020 Mar;8(2):442-449. doi: 10.1111/andr.12706. Epub 2019 Sep 29. Andrology. 2020. PMID: 31509646
-
Involvement of complexin 2 in docking, locking and unlocking of different SNARE complexes during sperm capacitation and induced acrosomal exocytosis.PLoS One. 2012;7(3):e32603. doi: 10.1371/journal.pone.0032603. Epub 2012 Mar 6. PLoS One. 2012. PMID: 22412896 Free PMC article.
-
Syntaxin and VAMP association with lipid rafts depends on cholesterol depletion in capacitating sperm cells.Mol Membr Biol. 2007 Jul-Aug;24(4):313-24. doi: 10.1080/09687860701228692. Mol Membr Biol. 2007. PMID: 17520487
-
Fusion of membranes during the acrosome reaction: a tale of two SNAREs.Mol Reprod Dev. 2000 Dec;57(4):309-10. doi: 10.1002/1098-2795(200012)57:4<309::AID-MRD1>3.0.CO;2-W. Mol Reprod Dev. 2000. PMID: 11066058 Review.
-
Capacitation-dependent reorganization of microdomains in the apical sperm head plasma membrane: functional relationship with zona binding and the zona-induced acrosome reaction.Theriogenology. 2008 Nov;70(8):1188-96. doi: 10.1016/j.theriogenology.2008.06.021. Epub 2008 Jul 21. Theriogenology. 2008. PMID: 18640708 Review.
Cited by
-
Snap29 mutant mice recapitulate neurological and ophthalmological abnormalities associated with 22q11 and CEDNIK syndrome.Commun Biol. 2019 Oct 11;2:375. doi: 10.1038/s42003-019-0601-5. eCollection 2019. Commun Biol. 2019. PMID: 31633066 Free PMC article.
-
Sperm Differentiation: The Role of Trafficking of Proteins.Int J Mol Sci. 2020 May 24;21(10):3702. doi: 10.3390/ijms21103702. Int J Mol Sci. 2020. PMID: 32456358 Free PMC article. Review.
References
-
- Salicioni AM, Platt MD, Wertheimer EV, Arcelay E, Allaire A, Sosnik J, Visconti PE. Signaling pathways involved in sperm capacitation. Soc Reprod Fertil Suppl, 2007: 65:245–259. - PubMed
-
- Abou-haila A, Tulsiani DRP. Signal transduction pathways that regulate sperm capacitation and the acrosome reaction. Arch Biochem Biophys, 2009, 485:72–81. - PubMed
-
- De Lamirande E., Capacitation as a regulatory event that primes spermatozoa for the acrosome reaction and fertilization. Mol Hum Reprod, 1997, 3:175–194. - PubMed
-
- Flesch FM, Brouwers JF, Nievelstein PF, Verkleij AJ, van Golde LM, Colenbrander B, Gadella BM. Bicarbonate stimulated phospholipid scrambling induces cholesterol redistribution and enables cholesterol depletion in the sperm plasma membrane. J Cell Sci, 2001, 114:3543–3555. - PubMed
-
- Nolan JP, Hammerstedt RH. Regulation of membrane stability and the acrosome reaction in mammalian sperm. FASEB J, 1997, 11:670–682. - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

