The improved efficacy of Sifuvirtide compared with enfuvirtide might be related to its selectivity for the rigid biomembrane, as determined through surface plasmon resonance
- PMID: 28207776
- PMCID: PMC5312942
- DOI: 10.1371/journal.pone.0171567
The improved efficacy of Sifuvirtide compared with enfuvirtide might be related to its selectivity for the rigid biomembrane, as determined through surface plasmon resonance
Abstract
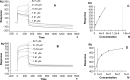
Most mechanistic studies on human immunodeficiency virus (HIV) peptide fusion inhibitors have focused on the interactions between fusion inhibitors and viral envelope proteins. However, the interactions of fusion inhibitors with viral membranes are also essential for the efficacy of these drugs. Here, we utilized surface plasmon resonance (SPR) technology to study the interactions between the HIV fusion inhibitor peptides sifuvirtide and enfuvirtide and biomembrane models. Sifuvirtide presented selectivity toward biomembrane models composed of saturated dipalmitoylphosphatidylcholine (DPPC) (32-fold higher compared with unsaturated 1-palmitoyl-2-oleoyl-sn-glycero-3-phosphocholine [POPC]) and sphingomyelin (SM) (31-fold higher compared with POPC), which are rigid compositions enriched in the HIV viral membrane. In contrast, enfuvirtide showed no significant selectively toward these rigid membrane models. Furthermore, the bindings of sifuvirtide and enfuvirtide to SM bilayers were markedly higher than those to monolayers (14-fold and 23-fold, respectively), indicating that the inner leaflet influences the binding of these drugs to SM bilayers. No obvious differences were noted in the bindings of either peptide to the other mono- and bilayer models tested, illustrating that both peptides interact with these membranes through surface-binding. The bindings of the inhibitor peptides to biomembranes were found to be driven predominantly by hydrophobic interactions rather than electrostatic interactions, as determined by comparing their affinities to those of positively charged 1-palmitoyl-2-oleoyl-sn-glycero-3-ethylphosphocholine (EPC) to zwitterionic membrane models. The improved efficiency of sifuvirtide relative to enfuvirtide might be related to its ability to adsorb on rigid lipidic areas, such as the viral envelope and lipid rafts, which results in an increased sifuvirtide concentration at the fusion site.
Conflict of interest statement
Figures

Similar articles
-
Sifuvirtide screens rigid membrane surfaces. establishment of a correlation between efficacy and membrane domain selectivity among HIV fusion inhibitor peptides.J Am Chem Soc. 2008 May 14;130(19):6215-23. doi: 10.1021/ja711247n. Epub 2008 Apr 15. J Am Chem Soc. 2008. PMID: 18410103
-
HIV fusion inhibitor peptide T-1249 is able to insert or adsorb to lipidic bilayers. Putative correlation with improved efficiency.J Am Chem Soc. 2004 Nov 17;126(45):14758-63. doi: 10.1021/ja0459882. J Am Chem Soc. 2004. PMID: 15535700
-
Synergistic efficacy of combination of enfuvirtide and sifuvirtide, the first- and next-generation HIV-fusion inhibitors.AIDS. 2009 Mar 13;23(5):639-41. doi: 10.1097/QAD.0b013e328325a4cd. AIDS. 2009. PMID: 19242316 Free PMC article.
-
[The current progress in the development of HIV-1 fusion inhibitors].Yao Xue Xue Bao. 2010 Feb;45(2):184-93. Yao Xue Xue Bao. 2010. PMID: 21351428 Review. Chinese.
-
Peptide inhibitors of virus-cell fusion: enfuvirtide as a case study in clinical discovery and development.Lancet Infect Dis. 2004 Jul;4(7):426-36. doi: 10.1016/S1473-3099(04)01058-8. Lancet Infect Dis. 2004. PMID: 15219553 Review.
Cited by
-
Peptides to combat viral infectious diseases.Peptides. 2020 Dec;134:170402. doi: 10.1016/j.peptides.2020.170402. Epub 2020 Sep 1. Peptides. 2020. PMID: 32889022 Free PMC article. Review.
-
Role of the Ebola membrane in the protection conferred by the three-mAb cocktail MIL77.Sci Rep. 2018 Dec 4;8(1):17628. doi: 10.1038/s41598-018-35964-6. Sci Rep. 2018. PMID: 30514891 Free PMC article.
-
The multifaceted nature of antimicrobial peptides: current synthetic chemistry approaches and future directions.Chem Soc Rev. 2021 Jul 5;50(13):7820-7880. doi: 10.1039/d0cs00729c. Chem Soc Rev. 2021. PMID: 34042120 Free PMC article. Review.
-
The application and prospects of antimicrobial peptides in antiviral therapy.Amino Acids. 2024 Dec 4;56(1):68. doi: 10.1007/s00726-024-03427-0. Amino Acids. 2024. PMID: 39630161 Free PMC article. Review.
-
Deep learning in the discovery of antiviral peptides and peptidomimetics: databases and prediction tools.Mol Divers. 2025 Aug;29(4):3753-3788. doi: 10.1007/s11030-025-11173-y. Epub 2025 Mar 28. Mol Divers. 2025. PMID: 40153158 Review.
References
-
- Baldwin CE, Sanders RW, Berkhout B. Inhibiting HIV-1 entry with fusion inhibitors. Curr Med Chem. 2003;10: 1633–1642. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources

