Deletion of junctional adhesion molecule A from platelets increases early-stage neointima formation after wire injury in hyperlipidemic mice
- PMID: 28211187
- PMCID: PMC5542900
- DOI: 10.1111/jcmm.13083
Deletion of junctional adhesion molecule A from platelets increases early-stage neointima formation after wire injury in hyperlipidemic mice
Abstract
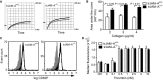
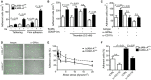
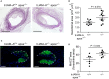
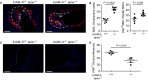
Platelets play an important role in the pathogenesis of vascular remodelling after injury. Junctional adhesion molecule A (JAM-A) was recently described to regulate platelet activation. Specific deletion of JAM-A from platelets resulted in increased reactivity and in accelerated progression of atherosclerosis. The aim of this study was to investigate the specific contribution of platelet-derived JAM-A to neointima formation after vascular injury. Mice with or without platelet-specific (tr)JAM-A-deficiency in an apolipoprotein e (apoe-/- ) background underwent wire-induced injury of the common carotid artery. Ex vivo imaging by two-photon microscopy revealed increased platelet coverage at the site of injury in trJAM-A-deficient mice. Cell recruitment assays showed increased adhesion of monocytic cells to activated JAM-A-deficient platelets than to control platelets. Inhibition of αM β2 or GPIbα, but not of CD62P, suppressed those differences. Up to 4 weeks after wire injury, intimal neoplasia and neointimal cellular content were analysed. Neointimal lesion area was increased in trJAM-A-/- apoe-/- mice and the lesions showed an increased macrophage accumulation and proliferating smooth muscle cells compared with trJAM-A+/+ apoe-/- littermates 2 weeks, but not 4 weeks after injury. Re-endothelialization was decreased in trJAM-A-/- apoe-/- mice compared with controls 2 weeks after injury, yet it was complete in both groups after 4 weeks. A platelet gain of function by deletion of JAM-A accelerates neointima formation only during earlier phases after vascular injury, through an increased recruitment of mononuclear cells. Thus, the contribution of platelets might become less important when neointima formation progresses to later stages.
Keywords: junctional adhesion molecule; leucocyte; neointima; platelet.
© 2017 The Authors. Journal of Cellular and Molecular Medicine published by John Wiley & Sons Ltd and Foundation for Cellular and Molecular Medicine.
Figures

Similar articles
-
Hyperreactivity of junctional adhesion molecule A-deficient platelets accelerates atherosclerosis in hyperlipidemic mice.Circ Res. 2015 Feb 13;116(4):587-99. doi: 10.1161/CIRCRESAHA.116.304035. Epub 2014 Dec 3. Circ Res. 2015. PMID: 25472975
-
CCL5 deficiency reduces neointima formation following arterial injury and thrombosis in apolipoprotein E-deficient mice.Thromb Res. 2016 Aug;144:136-43. doi: 10.1016/j.thromres.2016.06.013. Epub 2016 Jun 16. Thromb Res. 2016. PMID: 27337700
-
Importance of junctional adhesion molecule-A for neointimal lesion formation and infiltration in atherosclerosis-prone mice.Arterioscler Thromb Vasc Biol. 2006 Feb;26(2):e10-3. doi: 10.1161/01.ATV.0000197852.24529.4f. Epub 2005 Nov 23. Arterioscler Thromb Vasc Biol. 2006. PMID: 16306427
-
Inflammation and vascular injury: basic discovery to drug development.Circ J. 2012;76(8):1811-8. doi: 10.1253/circj.cj-12-0801. Epub 2012 Jul 30. Circ J. 2012. PMID: 22785436 Free PMC article. Review.
-
JAM-A: Adhesion Receptor and Signaling Regulator in Atherosclerosis.Curr Atheroscler Rep. 2025 Jul 29;27(1):75. doi: 10.1007/s11883-025-01322-x. Curr Atheroscler Rep. 2025. PMID: 40728654 Free PMC article. Review.
Cited by
-
Where the Action Is-Leukocyte Recruitment in Atherosclerosis.Front Cardiovasc Med. 2022 Jan 11;8:813984. doi: 10.3389/fcvm.2021.813984. eCollection 2021. Front Cardiovasc Med. 2022. PMID: 35087886 Free PMC article. Review.
-
The Roles of Junctional Adhesion Molecules (JAMs) in Cell Migration.Front Cell Dev Biol. 2022 Mar 9;10:843671. doi: 10.3389/fcell.2022.843671. eCollection 2022. Front Cell Dev Biol. 2022. PMID: 35356274 Free PMC article. Review.
-
Junctional adhesion molecule-A: functional diversity through molecular promiscuity.Cell Mol Life Sci. 2018 Apr;75(8):1393-1409. doi: 10.1007/s00018-017-2729-0. Epub 2017 Dec 14. Cell Mol Life Sci. 2018. PMID: 29238845 Free PMC article. Review.
-
Homophilic Interaction Between Transmembrane-JAM-A and Soluble JAM-A Regulates Thrombo-Inflammation: Implications for Coronary Artery Disease.JACC Basic Transl Sci. 2022 May 23;7(5):445-461. doi: 10.1016/j.jacbts.2022.03.003. eCollection 2022 May. JACC Basic Transl Sci. 2022. PMID: 35663628 Free PMC article.
-
LNCRNA OIP5-AS1 regulates oxidative low-density lipoprotein-mediated endothelial cell injury via miR-320a/LOX1 axis.Mol Cell Biochem. 2020 Apr;467(1-2):15-25. doi: 10.1007/s11010-020-03688-9. Epub 2020 Feb 18. Mol Cell Biochem. 2020. PMID: 32072428
References
-
- Weber C, Fraemohs L, Dejana E. The role of junctional adhesion molecules in vascular inflammation. Nat Rev Immunol. 2007; 7: 467–77. - PubMed
-
- Garrido‐Urbani S, Bradfield PF, Imhof BA. Tight junction dynamics: the role of junctional adhesion molecules (JAMs). Cell Tissue Res. 2014; 355: 701–15. - PubMed
-
- Nourshargh S, Krombach F, Dejana E. The role of JAM‐A and PECAM‐1 in modulating leukocyte infiltration in inflamed and ischemic tissues. J Leukoc Biol. 2006; 80: 714–8. - PubMed
-
- Ozaki H, Ishii K, Horiuchi H, et al Combined treatment of TNF‐alpha and IFN‐gamma causes redistribution of junctional adhesion molecule in human endothelial cells. J Immunol. 1999; 163: 553–7. - PubMed
-
- Ostermann G, Fraemohs L, Baltus T, et al Involvement of JAM‐A in mononuclear cell recruitment on inflamed or atherosclerotic endothelium: inhibition by soluble JAM‐A. Arterioscler Thromb Vasc Biol. 2005; 25: 729–35. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases
Miscellaneous

