Diversity and distribution of avian malaria and related haemosporidian parasites in captive birds from a Brazilian megalopolis
- PMID: 28212654
- PMCID: PMC5316177
- DOI: 10.1186/s12936-017-1729-8
Diversity and distribution of avian malaria and related haemosporidian parasites in captive birds from a Brazilian megalopolis
Abstract
Background: The role of zoos in conservation programmes has increased significantly in last decades, and the health of captive animals is essential to guarantee success of such programmes. However, zoo birds suffer from parasitic infections, which often are caused by malaria parasites and related haemosporidians. Studies determining the occurrence and diversity of these parasites, aiming better understanding infection influence on fitness of captive birds, are limited.
Methods: In 2011-2015, the prevalence and diversity of Plasmodium spp. and Haemoproteus spp. was examined in blood samples of 677 captive birds from the São Paulo Zoo, the largest zoo in Latin America. Molecular and microscopic diagnostic methods were used in parallel to detect and identify these infections.
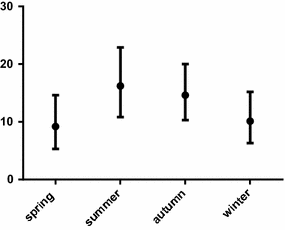
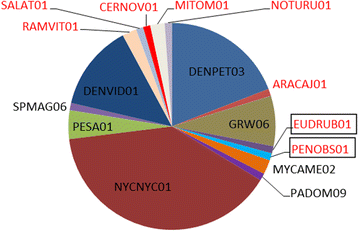
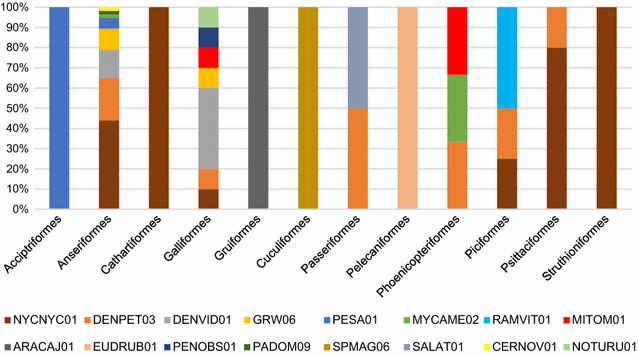
Results: The overall prevalence of haemosporidians was 12.6%. Parasites were mostly detected by the molecular diagnosis, indicating that many birds harbour subclinical or abortive infections. In this project, birds of 17 orders (almost half of all the orders currently accepted in taxonomy of birds), 29 families, and 122 species, were tested, detecting positive individuals in 27% of bird species. Birds from the Anatidae were the most prevalently infected (64.7% of all infected animals). In all, infections with parasites of the genus Plasmodium (overall prevalence 97.6%) predominated when compared to those of the genus Haemoproteus (2.4%). In total, 14 cytochrome b (cytb) lineages of Plasmodium spp. and 2 cytb lineages of Haemoproteus spp. were recorded. Eight lineages were new. One of the reported lineages was broad generalist while others were reported in single or a few species of birds. Molecular characterization of Haemoproteus ortalidum was developed.
Conclusion: This study shows that many species of birds are at risk in captivity. It is difficult to stop haemosporidian parasite transmission in zoos, but is possible to reduce the infection rate by treating the infected animals or/and while keeping them in facilities free from mosquitoes. Protocols of quarantine should be implemented whenever an animal is transferred between bird maintaining institutions. This is the first survey of haemosporidians in captive birds from different orders maintained in zoos. It is worth emphasizing the necessity of applying practices to control these parasites in management and husbandry of animals in captivity.
Keywords: Avian malaria; Captive birds; Conservation; Haemoproteus; Plasmodium; Zoo.
Figures






References
-
- Olney PJ, editor. Building a future for wildlife: the world zoo and aquarium conservation strategy. Bern: WAZA; 2005.
-
- EAZA. The Modern Zoo: Foundations for Management and Development. EAZA Executive Office. Amsterdam, the Netherlands. 2nd Edition; 2013.
-
- Atkinson CT, Thomas NJ, Hunter DB. Parasitic diseases of wild birds. Oxford: Wiley-Blackwell; 2008.
-
- Panayotova-Pencheva MS. Parasites in captive animals: a review of studies in some European zoos. Zool Garten. 2013;82:60–71. doi: 10.1016/j.zoolgart.2013.04.005. - DOI
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

