Activity-Induced Synaptic Structural Modifications by an Activator of Integrin Signaling at the Drosophila Neuromuscular Junction
- PMID: 28219985
- PMCID: PMC5373117
- DOI: 10.1523/JNEUROSCI.3128-16.2017
Activity-Induced Synaptic Structural Modifications by an Activator of Integrin Signaling at the Drosophila Neuromuscular Junction
Abstract
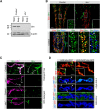
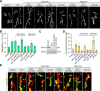
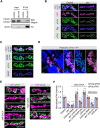
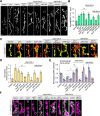
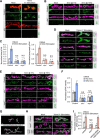
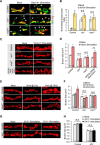
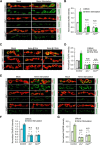
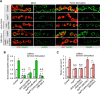
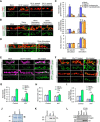
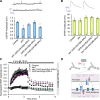
Activity-induced synaptic structural modification is crucial for neural development and synaptic plasticity, but the molecular players involved in this process are not well defined. Here, we report that a protein named Shriveled (Shv) regulates synaptic growth and activity-dependent synaptic remodeling at the Drosophila neuromuscular junction. Depletion of Shv causes synaptic overgrowth and an accumulation of immature boutons. We find that Shv physically and genetically interacts with βPS integrin. Furthermore, Shv is secreted during intense, but not mild, neuronal activity to acutely activate integrin signaling, induce synaptic bouton enlargement, and increase postsynaptic glutamate receptor abundance. Consequently, loss of Shv prevents activity-induced synapse maturation and abolishes post-tetanic potentiation, a form of synaptic plasticity. Our data identify Shv as a novel trans-synaptic signal secreted upon intense neuronal activity to promote synapse remodeling through integrin receptor signaling.SIGNIFICANCE STATEMENT The ability of neurons to rapidly modify synaptic structure in response to neuronal activity, a process called activity-induced structural remodeling, is crucial for neuronal development and complex brain functions. The molecular players that are important for this fundamental biological process are not well understood. Here we show that the Shriveled (Shv) protein is required during development to maintain normal synaptic growth. We further demonstrate that Shv is selectively released during intense neuronal activity, but not mild neuronal activity, to acutely activate integrin signaling and trigger structural modifications at the Drosophila neuromuscular junction. This work identifies Shv as a key modulator of activity-induced structural remodeling and suggests that neurons use distinct molecular cues to differentially modulate synaptic growth and remodeling to meet synaptic demand.
Keywords: Drosophila; Shriveled; activity-dependent structural remodeling; integrin; neuromuscular junction; synaptic boutons.
Copyright © 2017 the authors 0270-6474/17/373246-18$15.00/0.
Figures

References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
