Nucleobindin 1 binds to multiple types of pre-fibrillar amyloid and inhibits fibrillization
- PMID: 28220836
- PMCID: PMC5318909
- DOI: 10.1038/srep42880
Nucleobindin 1 binds to multiple types of pre-fibrillar amyloid and inhibits fibrillization
Abstract
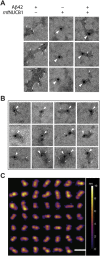
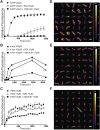
During amyloid fibril formation, amyloidogenic polypeptides misfold and self assemble into soluble pre-fibrillar aggregates, i.e., protofibrils, which elongate and mature into insoluble fibrillar aggregates. An emerging class of chaperones, chaperone-like amyloid binding proteins (CLABPs), has been shown to interfere with aggregation of particular misfolded amyloid peptides or proteins. We have discovered that the calcium-binding protein nuclebindin-1 (NUCB1) is a novel CLABP. We show that NUCB1 inhibits aggregation of islet-amyloid polypeptide associated with type 2 diabetes mellitus, a-synuclein associated with Parkinson's disease, transthyretin V30M mutant associated with familial amyloid polyneuropathy, and Aβ42 associated with Alzheimer's disease by stabilizing their respective protofibril intermediates. Kinetic studies employing the modeling software AmyloFit show that NUCB1 affects both primary nucleation and secondary nucleation. We hypothesize that NUCB1 binds to the common cross-β-sheet structure of protofibril aggregates to "cap" and stabilize soluble macromolecular complexes. Transmission electron microscopy and atomic force microscopy were employed to characterize the size, shape and volume distribution of multiple sources of NUCB1-capped protofibrils. Interestingly, NUCB1 prevents Aβ42 protofibril toxicity in a cellular assay. NUCB1-stabilized amyloid protofibrils could be used as immunogens to prepare conformation-specific antibodies and as novel tools to develop screens for anti-protofibril diagnostics and therapeutics.
Conflict of interest statement
The authors declare no competing financial interests.
Figures



Similar articles
-
Nucleobindin 1 caps human islet amyloid polypeptide protofibrils to prevent amyloid fibril formation.J Mol Biol. 2012 Aug 10;421(2-3):378-89. doi: 10.1016/j.jmb.2012.04.017. Epub 2012 Apr 24. J Mol Biol. 2012. PMID: 22542527 Free PMC article.
-
ABri peptide associated with familial British dementia forms annular and ring-like protofibrillar structures.Amyloid. 2004 Mar;11(1):10-3. doi: 10.1080/13506120410001667872. Amyloid. 2004. PMID: 15185493
-
Conformation-specific antibodies against multiple amyloid protofibril species from a single amyloid immunogen.J Cell Mol Med. 2019 Mar;23(3):2103-2114. doi: 10.1111/jcmm.14119. Epub 2019 Jan 20. J Cell Mol Med. 2019. PMID: 30663210 Free PMC article.
-
Protein denaturation and aggregation: Cellular responses to denatured and aggregated proteins.Ann N Y Acad Sci. 2005 Dec;1066:181-221. doi: 10.1196/annals.1363.030. Ann N Y Acad Sci. 2005. PMID: 16533927 Review.
-
Amyloid Oligomers: A Joint Experimental/Computational Perspective on Alzheimer's Disease, Parkinson's Disease, Type II Diabetes, and Amyotrophic Lateral Sclerosis.Chem Rev. 2021 Feb 24;121(4):2545-2647. doi: 10.1021/acs.chemrev.0c01122. Epub 2021 Feb 5. Chem Rev. 2021. PMID: 33543942 Free PMC article. Review.
Cited by
-
Neurodegeneration and humoral response proteins in cerebrospinal fluid associate with pediatric-onset multiple sclerosis and not monophasic demyelinating syndromes in childhood.Mult Scler. 2023 Jan;29(1):52-62. doi: 10.1177/13524585221125369. Epub 2022 Sep 24. Mult Scler. 2023. PMID: 36154753 Free PMC article.
-
Bri2 BRICHOS client specificity and chaperone activity are governed by assembly state.Nat Commun. 2017 Dec 12;8(1):2081. doi: 10.1038/s41467-017-02056-4. Nat Commun. 2017. PMID: 29234026 Free PMC article.
-
The ZiBuPiYin recipe regulates proteomic alterations in brain mitochondria-associated ER membranes caused by chronic psychological stress exposure: Implications for cognitive decline in Zucker diabetic fatty rats.Aging (Albany NY). 2020 Nov 18;12(23):23698-23726. doi: 10.18632/aging.103894. Epub 2020 Nov 18. Aging (Albany NY). 2020. PMID: 33221746 Free PMC article.
-
Fine mapping and identification of serum urate loci in American Indians: The Strong Heart Family Study.Sci Rep. 2019 Nov 29;9(1):17899. doi: 10.1038/s41598-019-52924-w. Sci Rep. 2019. PMID: 31784582 Free PMC article.
-
Chaperone-Mediated Responses and Mitochondrial-Endoplasmic Reticulum Coupling: Emerging Insight into Alzheimer's Disease.Cells. 2025 Jul 31;14(15):1179. doi: 10.3390/cells14151179. Cells. 2025. PMID: 40801612 Free PMC article. Review.
References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials
Miscellaneous

