Post translational modification of Parkin
- PMID: 28222786
- PMCID: PMC5319146
- DOI: 10.1186/s13062-017-0176-3
Post translational modification of Parkin
Abstract
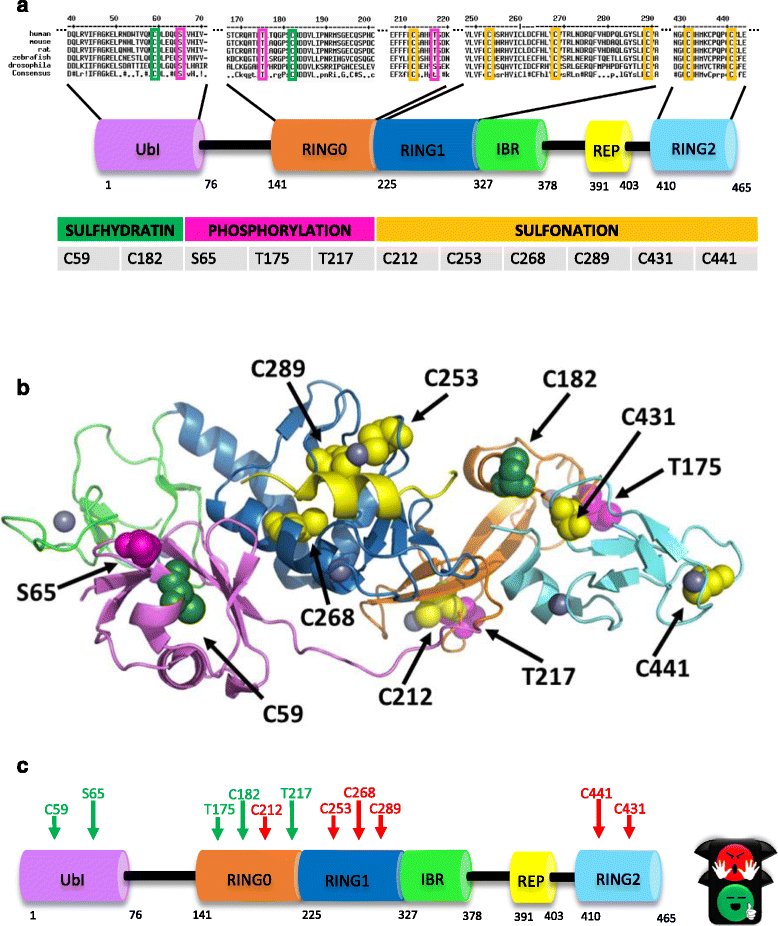
Mutations in the gene encoding for the E3 ubiquitin ligase Parkin are associated to a rare form of familiar autosomal recessive Parkinsonism. Despite decades of research on the Parkin protein, whose structure has been recently solved, little is known about the specific signalling pathways that lead to Parkin activation. Parkin activity spans from mitochondria quality control to tumor suppression and stress protection; it is thus tempting to hypothesize that the broad impact of Parkin on cellular physiology might be the result of different post translational modifications that can be controlled by balanced opposing events. Sequence alignment of Parkin from different species indicates high homology between domains across Parkin orthologs and identifies highly conserved amino acid residues that, if modified, impinge on Parkin functions. In this review, we summarize findings on post translational modifications that have been shown to affect Parkin activity and stability.
Reviewers: This article was reviewed by Prof. Dr. Konstanze F. Winklhofer and by Prof. Thomas Simmen. Both reviewers have been nominated by Professor Luca Pellegrini.
Keywords: Parkin; Parkinson’s disease; Phosphorylation; Post translational modifications; Ubiquitination.
Figures
Similar articles
-
Post-translational modification of Parkin and its research progress in cancer.Cancer Commun (Lond). 2019 Nov 21;39(1):77. doi: 10.1186/s40880-019-0421-5. Cancer Commun (Lond). 2019. PMID: 31753025 Free PMC article. Review.
-
Identification of a novel Zn2+-binding domain in the autosomal recessive juvenile Parkinson-related E3 ligase parkin.J Biol Chem. 2009 May 29;284(22):14978-86. doi: 10.1074/jbc.M808700200. Epub 2009 Apr 1. J Biol Chem. 2009. PMID: 19339245 Free PMC article.
-
The three 'P's of mitophagy: PARKIN, PINK1, and post-translational modifications.Genes Dev. 2015 May 15;29(10):989-99. doi: 10.1101/gad.262758.115. Genes Dev. 2015. PMID: 25995186 Free PMC article. Review.
-
Biochemical analysis of Parkinson's disease-causing variants of Parkin, an E3 ubiquitin-protein ligase with monoubiquitylation capacity.Hum Mol Genet. 2006 Jul 1;15(13):2059-75. doi: 10.1093/hmg/ddl131. Epub 2006 May 19. Hum Mol Genet. 2006. PMID: 16714300
-
Ubiquitin phosphorylated at Ser57 hyper-activates parkin.Biochim Biophys Acta Gen Subj. 2017 Nov;1861(11 Pt B):3038-3046. doi: 10.1016/j.bbagen.2017.06.023. Epub 2017 Jul 6. Biochim Biophys Acta Gen Subj. 2017. PMID: 28689991
Cited by
-
Post-translational modification of Parkin and its research progress in cancer.Cancer Commun (Lond). 2019 Nov 21;39(1):77. doi: 10.1186/s40880-019-0421-5. Cancer Commun (Lond). 2019. PMID: 31753025 Free PMC article. Review.
-
Mitochondrial Function and Parkinson's Disease: From the Perspective of the Electron Transport Chain.Front Mol Neurosci. 2021 Dec 9;14:797833. doi: 10.3389/fnmol.2021.797833. eCollection 2021. Front Mol Neurosci. 2021. PMID: 34955747 Free PMC article. Review.
-
A metabolomic signature of treated and drug-naïve patients with Parkinson's disease: a pilot study.Metabolomics. 2019 Jun 10;15(6):90. doi: 10.1007/s11306-019-1554-x. Metabolomics. 2019. PMID: 31183578
-
The impact of proteostasis dysfunction secondary to environmental and genetic causes on neurodegenerative diseases progression and potential therapeutic intervention.Environ Sci Pollut Res Int. 2020 Apr;27(11):11461-11483. doi: 10.1007/s11356-020-07914-1. Epub 2020 Feb 19. Environ Sci Pollut Res Int. 2020. PMID: 32072427 Review.
-
Age-associated insolubility of parkin in human midbrain is linked to redox balance and sequestration of reactive dopamine metabolites.Acta Neuropathol. 2021 May;141(5):725-754. doi: 10.1007/s00401-021-02285-4. Epub 2021 Mar 10. Acta Neuropathol. 2021. PMID: 33694021 Free PMC article.
References
-
- Thomas B, Beal MF. Parkinson’s disease. Human molecular genetics. 2007;16 Spec No. 2:R183-94. doi:10.1093/hmg/ddm159. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources

