Fe3O4-based PLGA nanoparticles as MR contrast agents for the detection of thrombosis
- PMID: 28223802
- PMCID: PMC5310639
- DOI: 10.2147/IJN.S123228
Fe3O4-based PLGA nanoparticles as MR contrast agents for the detection of thrombosis
Abstract
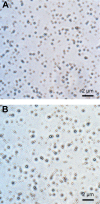
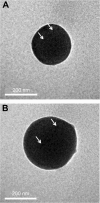
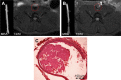
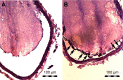
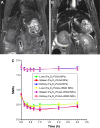
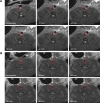
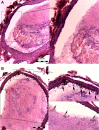
Thrombotic disease is a great threat to human health, and early detection is particularly important. Magnetic resonance (MR) molecular imaging provides noninvasive imaging with the potential for early disease diagnosis. In this study, we developed Fe3O4-based poly(lactic-co-glycolic acid) (PLGA) nanoparticles (NPs) surface-modified with a cyclic Arg-Gly-Asp (cRGD) peptide as an MR contrast agent for the detection of thrombosis. The physical and chemical characteristics, biological toxicity, ability to target thrombi, and biodistribution of the NPs were studied. The Fe3O4-PLGA-cRGD NPs were constructed successfully, and hematologic and pathologic assays indicated no in vivo toxicity of the NPs. In a rat model of FeCl3-induced abdominal aorta thrombosis, the NPs readily and selectively accumulated on the surface of the thrombosis and under vascular endothelial cells ex vivo and in vivo. In the in vivo experiment, the biodistribution of the NPs suggested that the NPs might be internalized by the macrophages of the reticuloendothelial system in the liver and the spleen. The T2 signal decreased at the mural thrombus 10 min after injection and then gradually increased until 50 min. These results suggest that the NPs are suitable for in vivo molecular imaging of thrombosis under high shear stress conditions and represent a very promising MR contrast agent for sensitive and specific detection of thrombosis.
Keywords: cyclic Arg-Gly-Asp peptide; iron oxide; magnetic resonance imaging; poly(lactic-co-glycolic acid); thrombosis.
Conflict of interest statement
Disclosure The authors report no conflicts of interest in this work.
Figures



References
-
- Whinna HC. Overview of murine thrombosis models. Thromb Res. 2008;122(Suppl 1):S64–S69. - PubMed
-
- Bender YY, Pfeifer A, Ebersberger HU, et al. Molecular cardiovascular magnetic resonance: current status and future prospects. Curr Cardiol Rep. 2016;18(5):47. - PubMed
-
- Wu W, Wang Y, Shen S, et al. In vivo ultrasound molecular imaging of inflammatory thrombosis in arteries with cyclic Arg-Gly-Asp-modified microbubbles targeted to glycoprotein IIb/IIIa. Invest Radiol. 2013;48(11):803–812. - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

