Epileptogenesis in organotypic hippocampal cultures has limited dependence on culture medium composition
- PMID: 28225808
- PMCID: PMC5321418
- DOI: 10.1371/journal.pone.0172677
Epileptogenesis in organotypic hippocampal cultures has limited dependence on culture medium composition
Abstract
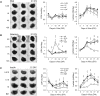
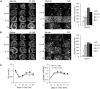
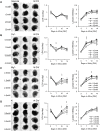
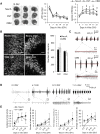
Rodent organotypic hippocampal cultures spontaneously develop epileptiform activity after approximately 2 weeks in vitro and are increasingly used as a model of chronic post-traumatic epilepsy. However, organotypic cultures are maintained in an artificial environment (culture medium), which contains electrolytes, glucose, amino acids and other components that are not present at the same concentrations in cerebrospinal fluid (CSF). Therefore, it is possible that epileptogenesis in organotypic cultures is driven by these components. We examined the influence of medium composition on epileptogenesis. Epileptogenesis was evaluated by measurements of lactate and lactate dehydrogenase (LDH) levels (biomarkers of ictal activity and cell death, respectively) in spent culture media, immunohistochemistry and automated 3-D cell counts, and extracellular recordings from CA3 regions. Changes in culture medium components moderately influenced lactate and LDH levels as well as electrographic seizure burden and cell death. However, epileptogenesis occurred in any culture medium that was capable of supporting neural survival. We conclude that medium composition is unlikely to be the cause of epileptogenesis in the organotypic hippocampal culture model of chronic post-traumatic epilepsy.
Conflict of interest statement
Figures




References
-
- McBain C, Boden P, Hill R. Rat hippocampal slices ‘in vitro’display spontaneous epileptiform activity following long-term organotypic culture. Journal of Neuroscience Methods. 1989;27(1):35–49. - PubMed
-
- Bausch SB, McNamara JO. Synaptic connections from multiple subfields contribute to granule cell hyperexcitability in hippocampal slice cultures. Journal of neurophysiology. 2000;84(6):2918–32. - PubMed
-
- Berdichevsky Y, Dryer AM, Saponjian Y, Mahoney MM, Pimentel CA, Lucini CA, et al. PI3K-Akt signaling activates mTOR-mediated epileptogenesis in organotypic hippocampal culture model of post-traumatic epilepsy. The Journal of Neuroscience. 2013;33(21):9056–67. 10.1523/JNEUROSCI.3870-12.2013 - DOI - PMC - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Miscellaneous

