Gel phase formation in dilute triblock copolyelectrolyte complexes
- PMID: 28230046
- PMCID: PMC5331217
- DOI: 10.1038/ncomms14131
Gel phase formation in dilute triblock copolyelectrolyte complexes
Abstract
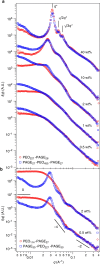
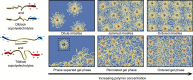
Assembly of oppositely charged triblock copolyelectrolytes into phase-separated gels at low polymer concentrations (<1% by mass) has been observed in scattering experiments and molecular dynamics simulations. Here we show that in contrast to uncharged, amphiphilic block copolymers that form discrete micelles at low concentrations and enter a phase of strongly interacting micelles in a gradual manner with increasing concentration, the formation of a dilute phase of individual micelles is prevented in polyelectrolyte complexation-driven assembly of triblock copolyelectrolytes. Gel phases form and phase separate almost instantaneously on solvation of the copolymers. Furthermore, molecular models of self-assembly demonstrate the presence of oligo-chain aggregates in early stages of copolyelectrolyte assembly, at experimentally unobservable polymer concentrations. Our discoveries contribute to the fundamental understanding of the structure and pathways of complexation-driven assemblies, and raise intriguing prospects for gel formation at extraordinarily low concentrations, with applications in tissue engineering, agriculture, water purification and theranostics.
Conflict of interest statement
The authors declare no competing financial interests.
Figures



References
-
- Harada A. & Kataoka K. Chain length recognition: core–shell supramolecular assembly from oppositely charged block copolymers. Science 283, 65–67 (1999). - PubMed
-
- Voets I. K., de Keizer A. & Cohen Stuart M. A. Complex coacervate core micelles. Adv. Colloid Interface Sci. 147, 300–318 (2009). - PubMed
-
- Pergushov D. V., Mueller A. H. E. & Schacher F. H. Micellar interpolyelectrolyte complexes. Chem. Soc. Rev. 41, 6888–6901 (2012). - PubMed
-
- Ou Z. & Muthukumar M. Entropy and enthalpy of polyelectrolyte complexation: Langevin dynamics simulations. J. Chem. Phys. 124, 154902 (2006). - PubMed
Publication types
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

