Flower-like superstructures of AIE-active tetraphenylethylene through solvophobic controlled self-assembly
- PMID: 28230060
- PMCID: PMC5322366
- DOI: 10.1038/srep42898
Flower-like superstructures of AIE-active tetraphenylethylene through solvophobic controlled self-assembly
Abstract
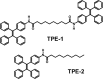
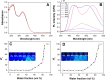
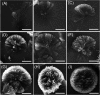
The development of well-organized structures with high luminescent properties in the solid and aggregated states is of both scientific and technological interest due to their applications in nanotechnology. In this paper we described the synthesis of amphiphilic and dumbbell shaped AIE-active tetraphenylethylene (TPE) derivatives and studied their self-assembly with solvophobic control. Interestingly, both TPE derivatives form a 3D flower-shape supramolecular structure from THF/water solutions at varying water fractions. SEM microscopy was used to visualise step-wise growth of flower-shape assembly. TPE derivatives also show good mechanochromic properties which can be observed in the process of grinding, fuming and heating. These TPE derivative self-assemblies are formed due to two main important properties: (i) the TPE-core along with alkyl chains, optimizing the dispersive interactions within a construct, and (ii) amide-linkage through molecular recognition. We believe such arrangements prevent crystallization and favour the directional growth of flower-shape nanostructures in a 3D fashion.
Conflict of interest statement
The authors declare no competing financial interests.
Figures






Similar articles
-
Flower-like supramolecular self-assembly of phosphonic acid appended naphthalene diimide and melamine.Sci Rep. 2015 Sep 29;5:14609. doi: 10.1038/srep14609. Sci Rep. 2015. PMID: 26416382 Free PMC article.
-
Right handed chiral superstructures from achiral molecules: self-assembly with a twist.Sci Rep. 2015 Oct 23;5:15652. doi: 10.1038/srep15652. Sci Rep. 2015. PMID: 26493294 Free PMC article.
-
Chiral Assembly of AIE-Active Achiral Molecules: An Odd Effect in Self-Assembly.Chemistry. 2017 Mar 17;23(16):3950-3956. doi: 10.1002/chem.201605458. Epub 2017 Feb 20. Chemistry. 2017. PMID: 28217844
-
Macrocycles and cages based on tetraphenylethylene with aggregation-induced emission effect.Chem Soc Rev. 2018 Oct 1;47(19):7452-7476. doi: 10.1039/c8cs00444g. Chem Soc Rev. 2018. PMID: 30177975 Review.
-
Multiple Anti-Counterfeiting Guarantees from a Simple Tetraphenylethylene Derivative - High-Contrasted and Multi-State Mechanochromism and Photochromism.Angew Chem Int Ed Engl. 2019 Dec 2;58(49):17814-17819. doi: 10.1002/anie.201910530. Epub 2019 Oct 22. Angew Chem Int Ed Engl. 2019. PMID: 31560154 Review.
Cited by
-
Thermo-Induced Fluorochromism in Two AIE Zinc Complexes: A Deep Insight into the Structure-Property Relationship.Molecules. 2022 Apr 14;27(8):2551. doi: 10.3390/molecules27082551. Molecules. 2022. PMID: 35458748 Free PMC article.
-
Self-assembled kanamycin antibiotic-inorganic microflowers and their application as a photocatalyst for the removal of organic dyes.Sci Rep. 2020 Jan 13;10(1):154. doi: 10.1038/s41598-019-57044-z. Sci Rep. 2020. PMID: 31932614 Free PMC article.
-
Co-Assembled Nanosystems Exhibiting Intrinsic Fluorescence by Complexation of Amino Terpolymer and Its Quaternized Analog with Aggregation-Induced Emission (AIE) Dye.Nanomaterials (Basel). 2024 Oct 11;14(20):1631. doi: 10.3390/nano14201631. Nanomaterials (Basel). 2024. PMID: 39452967 Free PMC article.
-
Fatty Acids/Tetraphenylethylene Conjugates: Hybrid AIEgens for the Preparation of Peptide-Based Supramolecular Gels.Front Chem. 2022 Aug 8;10:927563. doi: 10.3389/fchem.2022.927563. eCollection 2022. Front Chem. 2022. PMID: 36003614 Free PMC article.
-
Supramolecular organogels fabricated with dicarboxylic acids and primary alkyl amines: controllable self-assembled structures.RSC Adv. 2020 Aug 6;10(49):29129-29138. doi: 10.1039/d0ra05072e. eCollection 2020 Aug 5. RSC Adv. 2020. PMID: 35521101 Free PMC article.
References
-
- Lehn J. M. Supramolecular Chemistry: Concepts and Perspectives, VCH, Weinheim, Germany, 1995.
-
- Babu S. S., Praveen V. K. & Ajayaghosh A. Functional π-gelators and their applications. Chem. Rev. 114, 1973–2129 (2014). - PubMed
-
- Zhang X., Rehm S., Safont-Sempere M. M. & Würthner F. Vesicular perylene dye nanocapsules as supramolecular fluorescent pH sensor systems. Nat. Chem. 1, 623–629 (2009). - PubMed
Publication types
LinkOut - more resources
Full Text Sources
Other Literature Sources

