The moderating role of absorptive capacity and the differential effects of acquisitions and alliances on Big Pharma firms' innovation performance
- PMID: 28231332
- PMCID: PMC5322930
- DOI: 10.1371/journal.pone.0172488
The moderating role of absorptive capacity and the differential effects of acquisitions and alliances on Big Pharma firms' innovation performance
Abstract
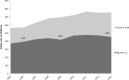
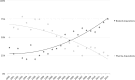
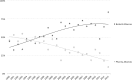
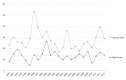
In the context of increased pharmaceutical innovation deficits and Big Pharma blockbusters' patent expirations, this paper examines the moderating role of firms' absorptive capacity in external innovation activities of Big Pharma firms. The study indicates a rising interest of Big Pharma in acquisitions of and alliances with biotechnology companies. Unfortunately, this increased interest is not reflected in the number of new drugs generated by Big Pharma. We find that acquisitions of biotech companies have negatively affected Big Pharma firms' innovation performance on average but these acquisitions might have a positive effect at higher levels of acquiring firms' absorptive capacity. Moreover, also acquisitions of pharma companies and alliances with biotech companies only have a positive effect on innovation performance at sufficiently high levels of absorptive capacity. The moderating role of absorptive capacity implicates that a tight integration of internal R&D efforts and (unrelated) external knowledge is crucial for harnessing complementarity effects.
Conflict of interest statement
Figures

References
-
- Gassmann O, Reepmeyer G. Organizing pharmaceutical innovation: from science‐based knowledge creators to drug‐oriented knowledge brokers. Creativity and Innovation Management. 2005;14(3):233–45.
MeSH terms
LinkOut - more resources
Full Text Sources
Other Literature Sources

