Ethylene production with engineered Synechocystis sp PCC 6803 strains
- PMID: 28231787
- PMCID: PMC5324202
- DOI: 10.1186/s12934-017-0645-5
Ethylene production with engineered Synechocystis sp PCC 6803 strains
Abstract
Background: Metabolic engineering and synthetic biology of cyanobacteria offer a promising sustainable alternative approach for fossil-based ethylene production, by using sunlight via oxygenic photosynthesis, to convert carbon dioxide directly into ethylene. Towards this, both well-studied cyanobacteria, i.e., Synechocystis sp PCC 6803 and Synechococcus elongatus PCC 7942, have been engineered to produce ethylene by introducing the ethylene-forming enzyme (Efe) from Pseudomonas syringae pv. phaseolicola PK2 (the Kudzu strain), which catalyzes the conversion of the ubiquitous tricarboxylic acid cycle intermediate 2-oxoglutarate into ethylene.
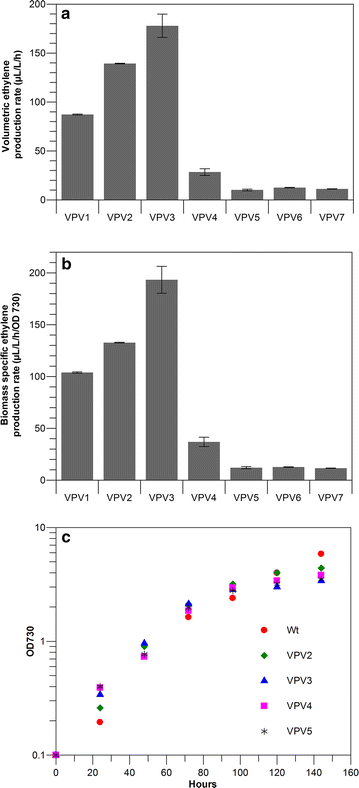
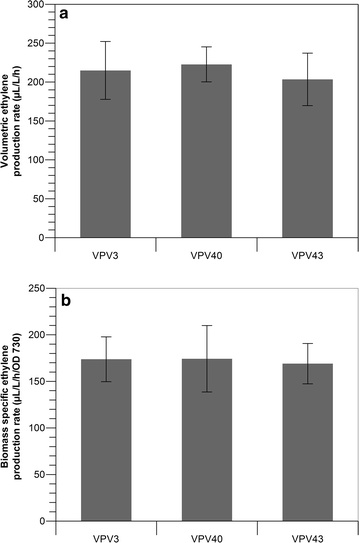
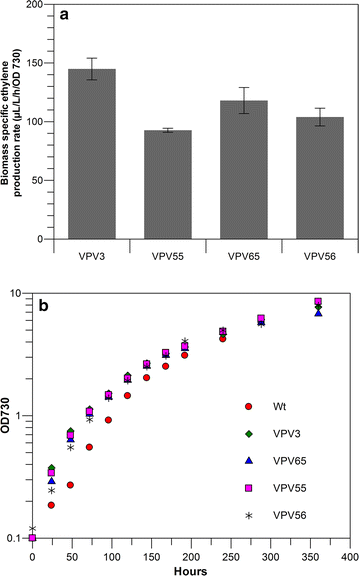
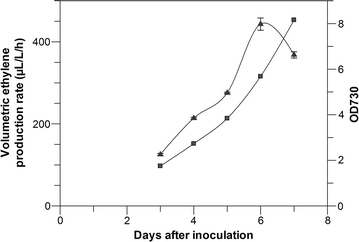
Results: This study focuses on Synechocystis sp PCC 6803 and shows stable ethylene production through the integration of a codon-optimized version of the efe gene under control of the Ptrc promoter and the core Shine-Dalgarno sequence (5'-AGGAGG-3') as the ribosome-binding site (RBS), at the slr0168 neutral site. We have increased ethylene production twofold by RBS screening and further investigated improving ethylene production from a single gene copy of efe, using multiple tandem promoters and by putting our best construct on an RSF1010-based broad-host-self-replicating plasmid, which has a higher copy number than the genome. Moreover, to raise the intracellular amounts of the key Efe substrate, 2-oxoglutarate, from which ethylene is formed, we constructed a glycogen-synthesis knockout mutant (ΔglgC) and introduced the ethylene biosynthetic pathway in it. Under nitrogen limiting conditions, the glycogen knockout strain has increased intracellular 2-oxoglutarate levels; however, surprisingly, ethylene production was lower in this strain than in the wild-type background.
Conclusion: Making use of different RBS sequences, production of ethylene ranging over a 20-fold difference has been achieved. However, a further increase of production through multiple tandem promoters and a broad-host plasmid was not achieved speculating that the transcription strength and the gene copy number are not the limiting factors in our system.
Keywords: Arginine; Cyanobacteria; Ethylene; Glycogen; Oxoglutarate; Sustainable; Synechocystis.
Figures

Similar articles
-
Enhanced stable production of ethylene in photosynthetic cyanobacterium Synechococcus elongatus PCC 7942.World J Microbiol Biotechnol. 2019 May 8;35(5):77. doi: 10.1007/s11274-019-2652-7. World J Microbiol Biotechnol. 2019. PMID: 31069553 Free PMC article.
-
Ethylene synthesis and regulated expression of recombinant protein in Synechocystis sp. PCC 6803.PLoS One. 2012;7(11):e50470. doi: 10.1371/journal.pone.0050470. Epub 2012 Nov 21. PLoS One. 2012. PMID: 23185630 Free PMC article.
-
A Genetic Toolbox for Modulating the Expression of Heterologous Genes in the Cyanobacterium Synechocystis sp. PCC 6803.ACS Synth Biol. 2018 Jan 19;7(1):276-286. doi: 10.1021/acssynbio.7b00297. Epub 2017 Dec 22. ACS Synth Biol. 2018. PMID: 29232504
-
Photoautotrophic production of renewable ethylene by engineered cyanobacteria: Steering the cell metabolism towards biotechnological use.Physiol Plant. 2021 Oct;173(2):579-590. doi: 10.1111/ppl.13430. Epub 2021 May 6. Physiol Plant. 2021. PMID: 33864400 Review.
-
[Direct biosynthesis of ethylene].Sheng Wu Gong Cheng Xue Bao. 2013 Oct;29(10):1431-40. Sheng Wu Gong Cheng Xue Bao. 2013. PMID: 24432658 Review. Chinese.
Cited by
-
Enhanced Production of High-Value Porphyrin Compound Heme by Metabolic Engineering Modification and Mixotrophic Cultivation of Synechocystis sp. PCC6803.Mar Drugs. 2024 Aug 23;22(9):378. doi: 10.3390/md22090378. Mar Drugs. 2024. PMID: 39330259 Free PMC article.
-
Lethality caused by ADP-glucose accumulation is suppressed by salt-induced carbon flux redirection in cyanobacteria.J Exp Bot. 2020 Mar 25;71(6):2005-2017. doi: 10.1093/jxb/erz559. J Exp Bot. 2020. PMID: 31858138 Free PMC article.
-
Bio-production of gaseous alkenes: ethylene, isoprene, isobutene.Biotechnol Biofuels. 2018 Aug 29;11:234. doi: 10.1186/s13068-018-1230-9. eCollection 2018. Biotechnol Biofuels. 2018. PMID: 30181774 Free PMC article. Review.
-
Structures and Mechanisms of the Non-Heme Fe(II)- and 2-Oxoglutarate-Dependent Ethylene-Forming Enzyme: Substrate Binding Creates a Twist.J Am Chem Soc. 2017 Aug 30;139(34):11980-11988. doi: 10.1021/jacs.7b06186. Epub 2017 Aug 22. J Am Chem Soc. 2017. PMID: 28780854 Free PMC article.
-
Light-Driven Synthetic Biology: Progress in Research and Industrialization of Cyanobacterial Cell Factory.Life (Basel). 2022 Oct 3;12(10):1537. doi: 10.3390/life12101537. Life (Basel). 2022. PMID: 36294972 Free PMC article. Review.
References
-
- Carrieri D, Paddock T, Maness PC, Seibert M, Yu J. Photo-catalytic conversion of carbon dioxide to organic acids by a recombinant cyanobacterium incapable of glycogen storage. Energy Environ Sci. 2012;5:9457–9461. doi: 10.1039/c2ee23181f. - DOI
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources

