Peripheral blood mononuclear cells from neovascular age-related macular degeneration patients produce higher levels of chemokines CCL2 (MCP-1) and CXCL8 (IL-8)
- PMID: 28231837
- PMCID: PMC5324243
- DOI: 10.1186/s12974-017-0820-y
Peripheral blood mononuclear cells from neovascular age-related macular degeneration patients produce higher levels of chemokines CCL2 (MCP-1) and CXCL8 (IL-8)
Abstract
Background: Infiltrating immune cells including monocytes/macrophages have been implicated in the pathogenesis of neovascular age-related macular degeneration (nAMD). The aim of this study was to investigate the cytokine and chemokine expression and secretion profile of peripheral blood mononuclear cells (PBMCs) from nAMD patients and the relationship between the cytokine/chemokine expression profile and clinical phenotype of nAMD, including macular fibrosis, macular atrophy or the responsiveness to anti-VEGF therapy.
Methods: One hundred sixty-one nAMD patients and 43 controls were enrolled in this study. nAMD patients were divided into subgroups based on the presence/absence of (1) macular atrophy, (2) macular fibrosis and (3) responsiveness to anti-VEGF therapy; 25-30 ml of peripheral blood were obtained from all participants and 5 ml were used for serum collection, and the remaining were used for PBMC isolation using density gradient centrifugation. Intracellular cytokine expressions by PBMCs following phorbol 12-myristate 13-acetate (PMA) and ionomycin stimulation were examined using flow cytometry. Cytokine productions in lipopolysaccharides (LPS)-or 1% oxygen -treated PBMC were measured using cytometric bead array (CBA) assay. In addition, cytokine and chemokine levels in the serum were also measured by CBA assay.
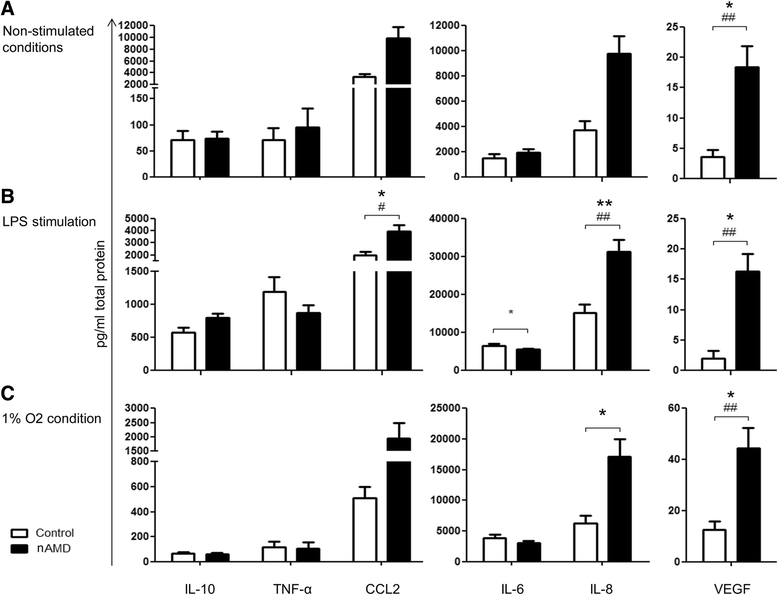
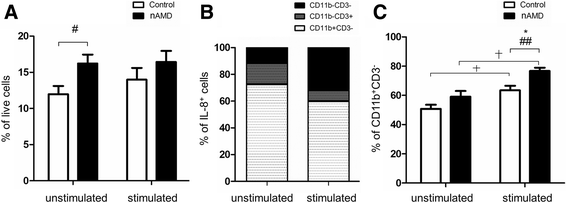
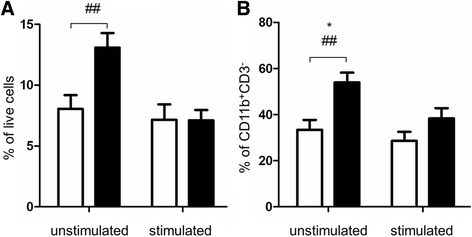
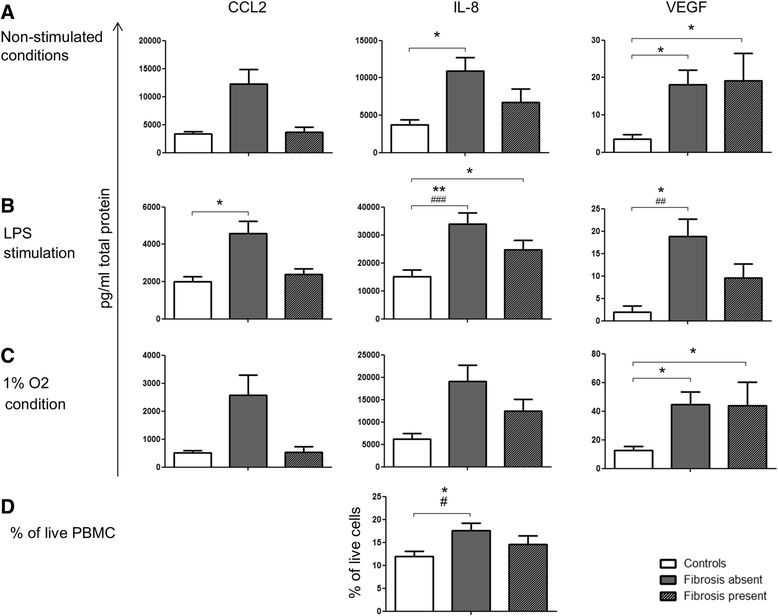
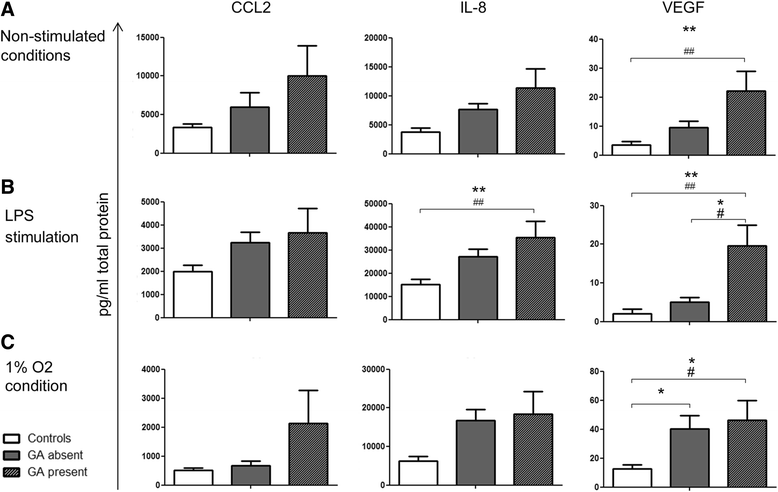
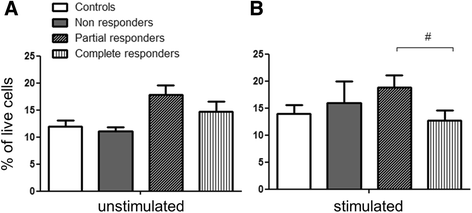
Results: PBMCs from nAMD patients secreted higher levels of IL-8, CCL2 and VEGF, especially following LPS and 1% oxygen stimulation, than those from controls. 60~80% of IL-8 producing cells were CD11b+CD3- monocytes. The percentage of CD11b+CD3- IL-8+ was significantly increased in nAMD patients compared to controls. PBMCs from nAMD patients without macular fibrosis produced the highest levels of IL-8 and CCL2, whilst PBMCs from nAMD patients with macular atrophy produced highest levels of VEGF. In addition, PBMCs from patients who partially responded to anti-VEGF produced higher levels of IL-8 compared to the cells from complete responders. Interestingly, serum level of CCL2 was not increased in nAMD patients although there was a trend of increased IL-8 in nAMD patients.
Conclusions: PBMCs, in particular monocytes, may contribute to CNV development in nAMD through secreting elevated levels of IL-8, CCL2 and VEGF after they are recruited to the macula. Apart from VEGF, IL-8 and CCL2 may be additional targets for nAMD management.
Keywords: Age-related macular degeneration; CCL2; Chemokines; Choroidal neovascularisation; Cytokines; Fibrosis; IL-8.
Figures
Similar articles
-
Inflammatory Factors of Macular Atrophy in Eyes With Neovascular Age-Related Macular Degeneration Treated With Aflibercept.Front Immunol. 2021 Oct 13;12:738521. doi: 10.3389/fimmu.2021.738521. eCollection 2021. Front Immunol. 2021. PMID: 34721402 Free PMC article.
-
Method development to quantify Bv8 expression in circulating CD11b+ cells in patients with neovascular age-related macular degeneration (nvAMD) exhibiting Anti-VEGF refractoriness.Exp Eye Res. 2016 Jul;148:45-51. doi: 10.1016/j.exer.2016.05.025. Epub 2016 May 30. Exp Eye Res. 2016. PMID: 27256991
-
T-helper-associated cytokines expression by peripheral blood mononuclear cells in patients with polypoidal choroidal vasculopathy and age-related macular degeneration.BMC Ophthalmol. 2016 Jun 7;16:80. doi: 10.1186/s12886-016-0251-z. BMC Ophthalmol. 2016. PMID: 27266510 Free PMC article.
-
Optimizing Anti-VEGF Treatment Outcomes for Patients with Neovascular Age-Related Macular Degeneration.J Manag Care Spec Pharm. 2018 Feb;24(2-a Suppl):S3-S15. doi: 10.18553/jmcp.2018.24.2-a.s3. J Manag Care Spec Pharm. 2018. PMID: 29383980 Free PMC article. Review.
-
Treatment Strategies for Anti-VEGF Resistance in Neovascular Age-Related Macular Degeneration by Targeting Arteriolar Choroidal Neovascularization.Biomolecules. 2024 Feb 21;14(3):252. doi: 10.3390/biom14030252. Biomolecules. 2024. PMID: 38540673 Free PMC article. Review.
Cited by
-
Quercetin Inhibits the Production of IL-1β-Induced Inflammatory Cytokines and Chemokines in ARPE-19 Cells via the MAPK and NF-κB Signaling Pathways.Int J Mol Sci. 2019 Jun 17;20(12):2957. doi: 10.3390/ijms20122957. Int J Mol Sci. 2019. PMID: 31212975 Free PMC article.
-
Association of CD11b+ Monocytes and Anti-Vascular Endothelial Growth Factor Injections in Treatment of Neovascular Age-Related Macular Degeneration and Polypoidal Choroidal Vasculopathy.JAMA Ophthalmol. 2019 May 1;137(5):515-522. doi: 10.1001/jamaophthalmol.2019.0010. JAMA Ophthalmol. 2019. PMID: 30844038 Free PMC article.
-
Immune Cells in Subretinal Wound Healing and Fibrosis.Front Cell Neurosci. 2022 Jun 10;16:916719. doi: 10.3389/fncel.2022.916719. eCollection 2022. Front Cell Neurosci. 2022. PMID: 35755781 Free PMC article. Review.
-
Protective Effects and Molecular Signaling of n-3 Fatty Acids on Oxidative Stress and Inflammation in Retinal Diseases.Antioxidants (Basel). 2020 Sep 26;9(10):920. doi: 10.3390/antiox9100920. Antioxidants (Basel). 2020. PMID: 32993153 Free PMC article. Review.
-
Glial-mediated neuroinflammatory mechanisms in age-related macular degeneration.Semin Immunopathol. 2022 Sep;44(5):673-683. doi: 10.1007/s00281-022-00939-3. Epub 2022 May 5. Semin Immunopathol. 2022. PMID: 35513496 Review.
References
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials
Miscellaneous

