GIGANTEA is a co-chaperone which facilitates maturation of ZEITLUPE in the Arabidopsis circadian clock
- PMID: 28232745
- PMCID: PMC5431898
- DOI: 10.1038/s41467-016-0014-9
GIGANTEA is a co-chaperone which facilitates maturation of ZEITLUPE in the Arabidopsis circadian clock
Abstract
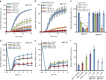
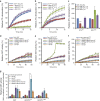
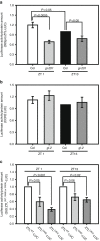
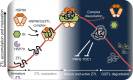
Circadian clock systems help establish the correct daily phasing of the behavioral, developmental, and molecular events needed for the proper coordination of physiology and metabolism. The circadian oscillator comprises transcription-translation feedback loops but also requires post-translational processes that regulate clock protein homeostasis. GIGANTEA is a unique plant protein involved in the maintenance and control of numerous facets of plant physiology and development. Through an unknown mechanism GIGANTEA stabilizes the F-box protein ZEITLUPE, a key regulator of the circadian clock. Here, we show that GIGANTEA has general protein chaperone activity and can act to specifically facilitate ZEITLUPE maturation into an active form in vitro and in planta. GIGANTEA forms a ternary complex with HSP90 and ZEITLUPE and its co-chaperone action synergistically enhances HSP90/HSP70 maturation of ZEITLUPE in vitro. These results identify a molecular mechanism for GIGANTEA activity that can explain its wide-ranging role in plant biology.The plant-specific GIGANTEA protein regulates the circadian clock by stabilizing the F-box protein ZEITLUPE via an unknown mechanism. Here Cha et al. show that GIGANTEA has intrinsic chaperone activity and can facilitate ZEITLUPE maturation by acting synergistically with HSP90.
Figures

Comment in
-
An HSP90 co-chaperone controls circadian proteostasis.Cell Cycle. 2017 Aug 18;16(16):1483-1484. doi: 10.1080/15384101.2017.1345238. Epub 2017 Jul 19. Cell Cycle. 2017. PMID: 28722533 Free PMC article. No abstract available.
References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

