Armed Oncolytic Adenovirus-Expressing PD-L1 Mini-Body Enhances Antitumor Effects of Chimeric Antigen Receptor T Cells in Solid Tumors
- PMID: 28235763
- PMCID: PMC5392365
- DOI: 10.1158/0008-5472.CAN-16-1577
Armed Oncolytic Adenovirus-Expressing PD-L1 Mini-Body Enhances Antitumor Effects of Chimeric Antigen Receptor T Cells in Solid Tumors
Abstract
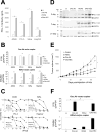
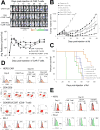
Chimeric antigen receptor-modified T cells (CAR T cells) produce proinflammatory cytokines that increase expression of T-cell checkpoint signals such as PD-L1, which may inhibit their functionality against solid tumors. In this study, we evaluated in human tumor xenograft models the proinflammatory properties of an oncolytic adenovirus (Onc.Ad) with a helper-dependent Ad (HDAd) that expresses a PD-L1 blocking mini-antibody (mini-body; HDPDL1) as a strategy to enhance CAR T-cell killing. Coadministration of these agents (CAd-VECPDL1) exhibited oncolytic effects with production of PD-L1 mini-body locally at the tumor site. On their own, HDPDL1 exhibited no antitumor effect and CAd-VECPDL1 alone reduced tumors only to volumes comparable to Onc.Ad treatment. However, combining CAd-VECPDL1 with HER2.CAR T cells enhanced antitumor activity compared with treatment with either HER2.CAR T cells alone or HER2.CAR T cells plus Onc.Ad. The benefits of locally produced PD-L1 mini-body by CAd-VECPDL1 could not be replicated by infusion of anti-PD-L1 IgG plus HER2.CAR T cells and coadministration of Onc.Ad in an HER2+ prostate cancer xenograft model. Overall, our data document the superiority of local production of PD-L1 mini-body by CAd-VECPDL1 combined with administration of tumor-directed CAR T cells to control the growth of solid tumors. Cancer Res; 77(8); 2040-51. ©2017 AACR.
©2017 American Association for Cancer Research.
Conflict of interest statement
Figures




Similar articles
-
Adenovirotherapy Delivering Cytokine and Checkpoint Inhibitor Augments CAR T Cells against Metastatic Head and Neck Cancer.Mol Ther. 2017 Nov 1;25(11):2440-2451. doi: 10.1016/j.ymthe.2017.09.010. Epub 2017 Sep 14. Mol Ther. 2017. PMID: 28974431 Free PMC article.
-
Mesenchymal stromal cell delivery of oncolytic immunotherapy improves CAR-T cell antitumor activity.Mol Ther. 2021 May 5;29(5):1808-1820. doi: 10.1016/j.ymthe.2021.02.004. Epub 2021 Feb 9. Mol Ther. 2021. PMID: 33571680 Free PMC article.
-
Oncolytic Adenovirus Armed with BiTE, Cytokine, and Checkpoint Inhibitor Enables CAR T Cells to Control the Growth of Heterogeneous Tumors.Mol Ther. 2020 May 6;28(5):1251-1262. doi: 10.1016/j.ymthe.2020.02.016. Epub 2020 Feb 24. Mol Ther. 2020. PMID: 32145203 Free PMC article.
-
Oncolytic Viruses Partner With T-Cell Therapy for Solid Tumor Treatment.Front Immunol. 2018 Sep 21;9:2103. doi: 10.3389/fimmu.2018.02103. eCollection 2018. Front Immunol. 2018. PMID: 30298067 Free PMC article. Review.
-
Recent advances in oncolytic adenovirus therapies for cancer.Curr Opin Virol. 2016 Dec;21:9-15. doi: 10.1016/j.coviro.2016.06.009. Epub 2016 Jul 2. Curr Opin Virol. 2016. PMID: 27379906 Free PMC article. Review.
Cited by
-
The Landscape of Nanovectors for Modulation in Cancer Immunotherapy.Pharmaceutics. 2022 Feb 11;14(2):397. doi: 10.3390/pharmaceutics14020397. Pharmaceutics. 2022. PMID: 35214129 Free PMC article. Review.
-
Improvement of the anticancer efficacy of PD-1/PD-L1 blockade via combination therapy and PD-L1 regulation.J Hematol Oncol. 2022 Mar 12;15(1):24. doi: 10.1186/s13045-022-01242-2. J Hematol Oncol. 2022. PMID: 35279217 Free PMC article. Review.
-
Insight into the Progress in CAR-T Cell Therapy and Combination with Other Therapies for Glioblastoma.Int J Gen Med. 2023 Sep 11;16:4121-4141. doi: 10.2147/IJGM.S418837. eCollection 2023. Int J Gen Med. 2023. PMID: 37720174 Free PMC article. Review.
-
The clinical study on treatment of CD19-directed chimeric antigen receptor-modified T cells in a case of refractory Richter syndrome.Cancer Med. 2019 Jun;8(6):2930-2941. doi: 10.1002/cam4.2193. Epub 2019 May 2. Cancer Med. 2019. PMID: 31050207 Free PMC article.
-
Overcoming Heterogeneity of Antigen Expression for Effective CAR T Cell Targeting of Cancers.Cancers (Basel). 2020 Apr 26;12(5):1075. doi: 10.3390/cancers12051075. Cancers (Basel). 2020. PMID: 32357417 Free PMC article. Review.
References
-
- Lichty BD, Breitbach CJ, Stojdl DF, Bell JC. Going viral with cancer immunotherapy. Nature reviews Cancer. 2014;14:559–67. - PubMed
-
- Cerullo V, Koski A, Vaha-Koskela M, Hemminki A. Chapter eight–Oncolytic adenoviruses for cancer immunotherapy: data from mice, hamsters, and humans. Advances in cancer research. 2012;115:265–318. - PubMed
-
- Kershaw MH, Westwood JA, Darcy PK. Gene-engineered T cells for cancer therapy. Nature reviews Cancer. 2013;13:525–41. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous

