Theiler's murine encephalomyelitis virus infection of SJL/J and C57BL/6J mice: Models for multiple sclerosis and epilepsy
- PMID: 28237622
- PMCID: PMC5474355
- DOI: 10.1016/j.jneuroim.2017.02.012
Theiler's murine encephalomyelitis virus infection of SJL/J and C57BL/6J mice: Models for multiple sclerosis and epilepsy
Abstract
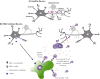
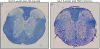
Mouse models are great tools to study the mechanisms of disease development. Theiler's murine encephalomyelitis virus is used in two distinct viral infection mouse models to study the human diseases multiple sclerosis (MS) and epilepsy. Intracerebral (i.c.) infection of the SJL/J mouse strain results in persistent viral infection of the central nervous system and a MS-like disease, while i.c. infection of the C57BL/6J mouse strain results in acute seizures and epilepsy. Our understanding of how the immune system contributes to the development of two disparate diseases caused by the same virus is presented.
Keywords: Epilepsy; Immune response; Mouse model; Multiple sclerosis (MS); Seizures; Theiler's murine encephalomyelitis virus (TMEV).
Copyright © 2017 Elsevier B.V. All rights reserved.
Figures

Similar articles
-
Viral mouse models of multiple sclerosis and epilepsy: Marked differences in neuropathogenesis following infection with two naturally occurring variants of Theiler's virus BeAn strain.Neurobiol Dis. 2017 Mar;99:121-132. doi: 10.1016/j.nbd.2016.12.020. Epub 2016 Dec 22. Neurobiol Dis. 2017. PMID: 28017800
-
Facets of Theiler's Murine Encephalomyelitis Virus-Induced Diseases: An Update.Int J Mol Sci. 2019 Jan 21;20(2):448. doi: 10.3390/ijms20020448. Int J Mol Sci. 2019. PMID: 30669615 Free PMC article. Review.
-
Two models of multiple sclerosis: experimental allergic encephalomyelitis (EAE) and Theiler's murine encephalomyelitis virus (TMEV) infection. A pathological and immunological comparison.Microsc Res Tech. 1995 Oct 15;32(3):215-29. doi: 10.1002/jemt.1070320305. Microsc Res Tech. 1995. PMID: 8527856 Free PMC article. Review.
-
Theiler's virus infection chronically alters seizure susceptibility.Epilepsia. 2010 Aug;51(8):1418-28. doi: 10.1111/j.1528-1167.2009.02405.x. Epub 2009 Dec 1. Epilepsia. 2010. PMID: 20002148
-
Brain inflammation, neurodegeneration and seizure development following picornavirus infection markedly differ among virus and mouse strains and substrains.Exp Neurol. 2016 May;279:57-74. doi: 10.1016/j.expneurol.2016.02.011. Epub 2016 Feb 15. Exp Neurol. 2016. PMID: 26892877
Cited by
-
How to Use the Cuprizone Model to Study De- and Remyelination.Int J Mol Sci. 2024 Jan 24;25(3):1445. doi: 10.3390/ijms25031445. Int J Mol Sci. 2024. PMID: 38338724 Free PMC article. Review.
-
The role of herpesvirus 6A and 6B in multiple sclerosis and epilepsy.Scand J Immunol. 2020 Dec;92(6):e12984. doi: 10.1111/sji.12984. Epub 2020 Oct 23. Scand J Immunol. 2020. PMID: 33037649 Free PMC article. Review.
-
Excessive Innate Immunity Steers Pathogenic Adaptive Immunity in the Development of Theiler's Virus-Induced Demyelinating Disease.Int J Mol Sci. 2021 May 17;22(10):5254. doi: 10.3390/ijms22105254. Int J Mol Sci. 2021. PMID: 34067536 Free PMC article. Review.
-
RNA-binding proteins as a common ground for neurodegeneration and inflammation in amyotrophic lateral sclerosis and multiple sclerosis.Front Mol Neurosci. 2023 Jul 4;16:1193636. doi: 10.3389/fnmol.2023.1193636. eCollection 2023. Front Mol Neurosci. 2023. PMID: 37475885 Free PMC article. Review.
-
The Development of Epilepsy Following CNS Viral Infections: Mechanisms.Curr Neurol Neurosci Rep. 2024 Nov 16;25(1):2. doi: 10.1007/s11910-024-01393-4. Curr Neurol Neurosci Rep. 2024. PMID: 39549124 Review.
References
-
- Annegers JF, Hauser WA, Beghi E, Nicolosi A, Kurland LT. The risk of unprovoked seizures after encephalitis and meningitis. Neurology. 1988;38:1407–1410. - PubMed
-
- Babbe H, Roers A, Waisman A, Lassmann H, Goebels N, Hohlfeld R, Friese M, Schroder R, Deckert M, Schmidt S, Ravid R, Rajewsky K. Clonal expansions of CD8(+) T cells dominate the T cell infiltrate in active multiple sclerosis lesions as shown by micromanipulation and single cell polymerase chain reaction. The Journal of experimental medicine. 2000;192:393–404. - PMC - PubMed
-
- Bale JF., Jr Virus and Immune-Mediated Encephalitides: Epidemiology, Diagnosis, Treatment, and Prevention. Pediatr Neurol. 2015;53:3–12. - PubMed
-
- Baranzini SE, Mudge J, van Velkinburgh JC, Khankhanian P, Khrebtukova I, Miller NA, Zhang L, Farmer AD, Bell CJ, Kim RW, May GD, Woodward JE, Caillier SJ, McElroy JP, Gomez R, Pando MJ, Clendenen LE, Ganusova EE, Schilkey FD, Ramaraj T, Khan OA, Huntley JJ, Luo S, Kwok PY, Wu TD, Schroth GP, Oksenberg JR, Hauser SL, Kingsmore SF. Genome, epigenome and RNA sequences of monozygotic twins discordant for multiple sclerosis. Nature. 2010;464:1351–1356. - PMC - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

