Self-Administration of Smokeless Tobacco Products in Rats
- PMID: 28239627
- PMCID: PMC5321097
- DOI: 10.18001/TRS.2.4.5
Self-Administration of Smokeless Tobacco Products in Rats
Abstract
Objective: Preclinical abuse liability assessment is an essential component of tobacco regulatory science. The goal of this project was to evaluate the relative abuse liability of smokeless tobacco products in rats using aqueous extracts of those products. These extracts provide exposure to an extensive range of nicotine and non-nicotine tobacco constituents as occurs in humans.
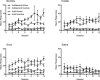
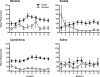
Methods: Rats were trained to self-administer either nicotine alone or extracts of Camel Snus or Kodiak smokeless tobacco at an equivalent nicotine unit dose. In Experiment 1, the relative reinforcing efficacy of these formulations was assessed in adults and adolescents using a progressive ratio schedule under limited-access conditions. In Experiment 2, relative reinforcing efficacy was assessed in adolescents under unlimited-access conditions using behavioral economic demand curve analysis.
Results: The reinforcing efficacy of nicotine formulations was higher in adolescents than adults, but no difference was observed between formulations in either age group. Similarly, there was no difference in elasticity of demand between formulations in adolescents.
Conclusions: The present findings suggest that the abuse liability of these smokeless tobacco products is similar to nicotine alone, and that nicotine dose is the primary determinant of the reinforcing efficacy of systemic exposure to these products.
Keywords: abuse liability; behavioral economics; nicotine; progressive-ratio schedule; rat; self-administration; smokeless tobacco extracts.
Figures
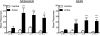
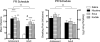

Similar articles
-
Animal models to assess the abuse liability of tobacco products: effects of smokeless tobacco extracts on intracranial self-stimulation.Drug Alcohol Depend. 2015 Feb 1;147:60-7. doi: 10.1016/j.drugalcdep.2014.12.015. Epub 2014 Dec 23. Drug Alcohol Depend. 2015. PMID: 25561387 Free PMC article.
-
Naturalistic assessment of demand for cigarettes, snus, and nicotine gum.Psychopharmacology (Berl). 2017 Jan;234(2):245-254. doi: 10.1007/s00213-016-4455-y. Epub 2016 Oct 11. Psychopharmacology (Berl). 2017. PMID: 27730273 Free PMC article.
-
Abuse liability assessment of an e-cigarette refill liquid using intracranial self-stimulation and self-administration models in rats.Drug Alcohol Depend. 2016 Nov 1;168:76-88. doi: 10.1016/j.drugalcdep.2016.08.628. Epub 2016 Sep 1. Drug Alcohol Depend. 2016. PMID: 27627814 Free PMC article.
-
Smokeless tobacco: an addicting drug.Adv Dent Res. 1997 Sep;11(3):330-5. doi: 10.1177/08959374970110030401. Adv Dent Res. 1997. PMID: 9524433 Review.
-
The Reinforcing Effects of Nicotine in Humans and Nonhuman Primates: A Review of Intravenous Self-Administration Evidence and Future Directions for Research.Nicotine Tob Res. 2015 Nov;17(11):1297-310. doi: 10.1093/ntr/ntv002. Epub 2015 Feb 10. Nicotine Tob Res. 2015. PMID: 25673111 Review.
Cited by
-
Advances in Behavioral Laboratory Methods that Inform Tobacco Regulatory Science: A TCORS Working Group Special Issue.Tob Regul Sci. 2016 Oct;2(4):294-300. doi: 10.18001/TRS.2.4.1. Epub 2016 Oct 1. Tob Regul Sci. 2016. PMID: 29152546 Free PMC article.
-
Attenuating nicotine's effects with high affinity human anti-nicotine monoclonal antibodies.PLoS One. 2021 Jul 30;16(7):e0254247. doi: 10.1371/journal.pone.0254247. eCollection 2021. PLoS One. 2021. PMID: 34329335 Free PMC article.
-
Non-nicotine constituents in e-cigarette aerosol extract attenuate nicotine's aversive effects in adolescent rats.Drug Alcohol Depend. 2019 Oct 1;203:51-60. doi: 10.1016/j.drugalcdep.2019.05.023. Epub 2019 Aug 1. Drug Alcohol Depend. 2019. PMID: 31404849 Free PMC article.
-
The nicotine-degrading enzyme NicA2 reduces nicotine levels in blood, nicotine distribution to brain, and nicotine discrimination and reinforcement in rats.BMC Biotechnol. 2018 Jul 24;18(1):46. doi: 10.1186/s12896-018-0457-7. BMC Biotechnol. 2018. PMID: 30041697 Free PMC article.
-
The role of β-Nicotyrine in E-Cigarette abuse liability I: Drug Discrimination.bioRxiv [Preprint]. 2024 Jul 16:2024.07.12.603310. doi: 10.1101/2024.07.12.603310. bioRxiv. 2024. PMID: 39071347 Free PMC article. Preprint.
References
-
- Belluzzi JD, Wang R, Leslie FM. Acetaldehyde enhances acquisition of nicotine self-administration in adolescent rats. Neuropsychopharmacology. 2005;30(4):705–712. doi:10.1038/sj.npp.1300586. - PubMed
-
- Clemens K, Caillé S, Stinus L, Cador M. The addition of five minor tobacco alkaloids increases nicotine-induced hyperactivity, sensitization and intravenous self-administration in rats. Int J Neuropsychopharmacol. 2009;12(10):1–12. doi:10.1017/S1461145709000273. - PubMed
-
- Brennan KA, Laugesen M, Truman P. Whole tobacco smoke extracts to model tobacco dependence in animals. Neuroscience and biobehavioral reviews. 2014;47:53–69. doi:10.1016/j.neubiorev.2014.07.014. - PubMed
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
