Serelaxin as a potential treatment for renal dysfunction in cirrhosis: Preclinical evaluation and results of a randomized phase 2 trial
- PMID: 28245243
- PMCID: PMC5330452
- DOI: 10.1371/journal.pmed.1002248
Serelaxin as a potential treatment for renal dysfunction in cirrhosis: Preclinical evaluation and results of a randomized phase 2 trial
Abstract
Background: Chronic liver scarring from any cause leads to cirrhosis, portal hypertension, and a progressive decline in renal blood flow and renal function. Extreme renal vasoconstriction characterizes hepatorenal syndrome, a functional and potentially reversible form of acute kidney injury in patients with advanced cirrhosis, but current therapy with systemic vasoconstrictors is ineffective in a substantial proportion of patients and is limited by ischemic adverse events. Serelaxin (recombinant human relaxin-2) is a peptide molecule with anti-fibrotic and vasoprotective properties that binds to relaxin family peptide receptor-1 (RXFP1) and has been shown to increase renal perfusion in healthy human volunteers. We hypothesized that serelaxin could ameliorate renal vasoconstriction and renal dysfunction in patients with cirrhosis and portal hypertension.
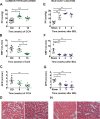
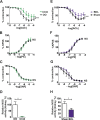
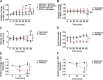
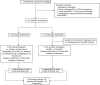
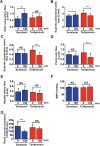
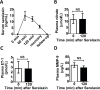
Methods and findings: To establish preclinical proof of concept, we developed two independent rat models of cirrhosis that were characterized by progressive reduction in renal blood flow and glomerular filtration rate and showed evidence of renal endothelial dysfunction. We then set out to further explore and validate our hypothesis in a phase 2 randomized open-label parallel-group study in male and female patients with alcohol-related cirrhosis and portal hypertension. Forty patients were randomized 1:1 to treatment with serelaxin intravenous (i.v.) infusion (for 60 min at 80 μg/kg/d and then 60 min at 30 μg/kg/d) or terlipressin (single 2-mg i.v. bolus), and the regional hemodynamic effects were quantified by phase contrast magnetic resonance angiography at baseline and after 120 min. The primary endpoint was the change from baseline in total renal artery blood flow. Therapeutic targeting of renal vasoconstriction with serelaxin in the rat models increased kidney perfusion, oxygenation, and function through reduction in renal vascular resistance, reversal of endothelial dysfunction, and increased activation of the AKT/eNOS/NO signaling pathway in the kidney. In the randomized clinical study, infusion of serelaxin for 120 min increased total renal arterial blood flow by 65% (95% CI 40%, 95%; p < 0.001) from baseline. Administration of serelaxin was safe and well tolerated, with no detrimental effect on systemic blood pressure or hepatic perfusion. The clinical study's main limitations were the relatively small sample size and stable, well-compensated population.
Conclusions: Our mechanistic findings in rat models and exploratory study in human cirrhosis suggest the therapeutic potential of selective renal vasodilation using serelaxin as a new treatment for renal dysfunction in cirrhosis, although further validation in patients with more advanced cirrhosis and renal dysfunction is required.
Trial registration: ClinicalTrials.gov NCT01640964.
Conflict of interest statement
PCH has received consultancy fees from Novartis and GORE. JAF has received consultancy fees from Merck and Novartis and research grant funding from GlaxoSmithKline. TS, JP, DY and ND are currently employees of Novartis and own either Novartis stocks or shares. TS, RS, JP, DY, and ND contributed to the Phase 2 trial of Serelaxin as Novartis associates but played no role in the independent preclinical research. RS is currently an employee of GlaxoSmithKline. DJW is a member of a Data Safety Monitoring Board for AbbVie for a phase 3 trial with atrasentan in renal disease (institutional) and a non-executive Board member of the Medicines and Healthcare products Regulatory Agency. DJW's research is supported by BHF, MRC and Wellcome Trust. JSD owns Biogen, Vertex, Regulus and BMS stock. JSD has patents for therapies to treat acute kidney injury.
Figures


References
-
- Besso J, Pru C, Padron J, Plaz J. Hepatorenal syndrome In: Gullo A, Lumb D, editors. Intensive and critical care medicine: reflections, recommendations and perspectives. Milan: Springer-Verlag; 2007. pp. 9–27.
Publication types
MeSH terms
Substances
Associated data
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

