Glutathione Peroxidase-Like Activity of Amino-Substituted Water-Soluble Cyclic Selenides: A Shift of the Major Catalytic Cycle in Methanol
- PMID: 28245615
- PMCID: PMC6155421
- DOI: 10.3390/molecules22030354
Glutathione Peroxidase-Like Activity of Amino-Substituted Water-Soluble Cyclic Selenides: A Shift of the Major Catalytic Cycle in Methanol
Abstract
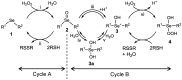
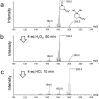
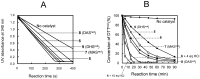
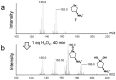
We previously reported that water-soluble cyclic selenides can mimic the antioxidative function of glutathione peroxidase (GPx) in water through a simple catalytic cycle, in which the selenide (>Se) is oxidized by H₂O₂ to the selenoxide (>Se=O) and the selenoxide is reduced by a thiol back to the selenide. In methanol, however, the GPx-like activity could not be explained by this simple scenario. To look into the reasons for the unusual behaviors in methanol, monoamino-substituted cyclic selenides with a variable ring size were synthesized, and the intermediates of the catalytic cycle were characterized by means of 77Se-NMR and LC-MS spectroscopies. In water, it was confirmed that the selenide and the selenoxide mainly contribute to the antioxidative function, though a slight contribution from the dihydroxy selenane (>Se(OH)₂) was also suggested. In methanol, on the other hand, other active species, such as hydroxyselenonium (>Se⁺-OH) and hydroxy perhydroxy selenane (>Se(OH)(OOH)), could be generated to build another catalytic cycle. This over-oxidation would be more feasible for amino-substituted cyclic selenides, probably because the ammonium (NH₃⁺) group would transfer a proton to the selenoxide moiety to produce a hydroxyselenonium species in the absence of an additional proton source. Thus, a shift of the major catalytic cycle in methanol would make the GPx-like antioxidative function of selenides perplexing.
Keywords: antioxidant; enzyme model; glutathione peroxidase; hydroxy perhydroxy selenane; selenide; selenoxide.
Conflict of interest statement
The authors declare no conflict of interest. The founding sponsors had no role in the design of the study; in the collection, analyses, or interpretation of data; in the writing of the manuscript, and in the decision to publish the results.
Figures
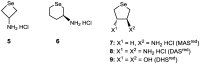
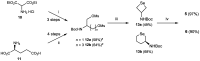
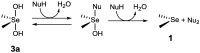

Similar articles
-
GPx-Like activity of selenides and selenoxides: experimental evidence for the involvement of hydroxy perhydroxy selenane as the active species.J Am Chem Soc. 2012 Jan 11;134(1):138-41. doi: 10.1021/ja209570y. Epub 2011 Dec 9. J Am Chem Soc. 2012. PMID: 22136421
-
Effects of ring size and polar functional groups on the glutathione peroxidase-like antioxidant activity of water-soluble cyclic selenides.J Org Chem. 2015 Jun 5;80(11):5633-42. doi: 10.1021/acs.joc.5b00544. Epub 2015 May 14. J Org Chem. 2015. PMID: 25941977
-
Diselenides and allyl selenides as glutathione peroxidase mimetics. Remarkable activity of cyclic seleninates produced in situ by the oxidation of allyl omega-hydroxyalkyl selenides.J Am Chem Soc. 2003 Nov 5;125(44):13455-60. doi: 10.1021/ja0357588. J Am Chem Soc. 2003. PMID: 14583041
-
Functional mimics of glutathione peroxidase: bioinspired synthetic antioxidants.Acc Chem Res. 2010 Nov 16;43(11):1408-19. doi: 10.1021/ar100059g. Epub 2010 Aug 6. Acc Chem Res. 2010. PMID: 20690615 Review.
-
Cyclic Seleninate Esters, Spirodioxyselenuranes and Related Compounds: New Classes of Biological Antioxidants That Emulate Glutathione Peroxidase.Chemistry. 2018 Jul 11;24(39):9714-9728. doi: 10.1002/chem.201800182. Epub 2018 Apr 30. Chemistry. 2018. PMID: 29542192 Review.
Cited by
-
Celebrating Two Centuries of Research in Selenium Chemistry: State of the Art and New Prospective.Molecules. 2017 Dec 2;22(12):2124. doi: 10.3390/molecules22122124. Molecules. 2017. PMID: 29207462 Free PMC article.
-
Exploring the Chemistry and Applications of Thio-, Seleno-, and Tellurosugars.Molecules. 2025 May 5;30(9):2053. doi: 10.3390/molecules30092053. Molecules. 2025. PMID: 40363858 Free PMC article. Review.
References
-
- Hepel M., Andreescu S. Oxidative Stress: Diagnostics, Prevention, and Therapy Volume 2. American Chemical Society; Washington, DC, USA: 2015. Oxidative stress and human health; pp. 1–33. (ACS Symposium Series).
-
- Alberto E.E., do Nascimento V., Braga A.L. Catalytic application of selenium and tellurium compounds as glutathione peroxidase enzyme mimetics. J. Braz. Chem. Soc. 2010;21:2032–2041. doi: 10.1590/S0103-50532010001100004. - DOI
-
- Santi C., Tidei C., Scalera C., Piroddi M., Galli F. Selenium Containing Compounds from Poison to Drug Candidates: A Review on the GPx-like Activity. Curr. Chem. Biol. 2013;7:25–36. doi: 10.2174/2212796811307010003. - DOI
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources

