Dgat1 and Dgat2 regulate enterocyte triacylglycerol distribution and alter proteins associated with cytoplasmic lipid droplets in response to dietary fat
- PMID: 28249764
- PMCID: PMC5503214
- DOI: 10.1016/j.bbalip.2017.02.014
Dgat1 and Dgat2 regulate enterocyte triacylglycerol distribution and alter proteins associated with cytoplasmic lipid droplets in response to dietary fat
Abstract
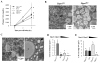
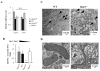
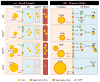
Enterocytes, the absorptive cells of the small intestine, mediate efficient absorption of dietary fat (triacylglycerol, TAG). The digestive products of dietary fat are taken up by enterocytes, re-esterified into TAG, and packaged on chylomicrons (CMs) for secretion into blood or temporarily stored within cytoplasmic lipid droplets (CLDs). Altered enterocyte TAG distribution impacts susceptibility to high fat diet associated diseases, but molecular mechanisms directing TAG toward these fates are unclear. Two enzymes, acyl CoA: diacylglycerol acyltransferase 1 (Dgat1) and Dgat2, catalyze the final, committed step of TAG synthesis within enterocytes. Mice with intestine-specific overexpression of Dgat1 (Dgat1Int) or Dgat2 (Dgat2Int), or lack of Dgat1 (Dgat1-/-), were previously found to have altered intestinal TAG secretion and storage. We hypothesized that varying intestinal Dgat1 and Dgat2 levels alters TAG distribution in subcellular pools for CM synthesis as well as the morphology and proteome of CLDs. To test this we used ultrastructural and proteomic methods to investigate intracellular TAG distribution and CLD-associated proteins in enterocytes from Dgat1Int, Dgat2Int, and Dgat1-/- mice 2h after a 200μl oral olive oil gavage. We found that varying levels of intestinal Dgat1 and Dgat2 altered TAG pools involved in CM assembly and secretion, the number or size of CLDs present in enterocytes, and the enterocyte CLD proteome. Overall, these results support a model where Dgat1 and Dgat2 function coordinately to regulate the process of dietary fat absorption by preferentially synthesizing TAG for incorporation into distinct subcellular TAG pools in enterocytes.
Keywords: Chylomicron; Cytoplasmic lipid droplet; DGAT; Enterocyte; Triacylglycerol.
Copyright © 2017 Elsevier B.V. All rights reserved.
Figures




Similar articles
-
DGAT1 deficiency disrupts lysosome function in enterocytes during dietary fat absorption.Biochim Biophys Acta Mol Cell Biol Lipids. 2019 Apr;1864(4):587-595. doi: 10.1016/j.bbalip.2018.10.007. Epub 2018 Oct 17. Biochim Biophys Acta Mol Cell Biol Lipids. 2019. PMID: 30342099 Free PMC article.
-
DGAT1 is not essential for intestinal triacylglycerol absorption or chylomicron synthesis.J Biol Chem. 2002 Jul 12;277(28):25474-9. doi: 10.1074/jbc.M202013200. Epub 2002 Apr 16. J Biol Chem. 2002. PMID: 11959864
-
Characterization of cytoplasmic lipid droplets in each region of the small intestine of lean and diet-induced obese mice in response to dietary fat.Am J Physiol Gastrointest Liver Physiol. 2021 Jul 1;321(1):G75-G86. doi: 10.1152/ajpgi.00084.2021. Epub 2021 May 19. Am J Physiol Gastrointest Liver Physiol. 2021. PMID: 34009042 Free PMC article.
-
Recent discoveries on absorption of dietary fat: Presence, synthesis, and metabolism of cytoplasmic lipid droplets within enterocytes.Biochim Biophys Acta. 2016 Aug;1861(8 Pt A):730-47. doi: 10.1016/j.bbalip.2016.04.012. Epub 2016 Apr 20. Biochim Biophys Acta. 2016. PMID: 27108063 Free PMC article. Review.
-
Role of DGAT enzymes in triacylglycerol metabolism.Arch Biochem Biophys. 2018 Oct 1;655:1-11. doi: 10.1016/j.abb.2018.08.001. Epub 2018 Aug 3. Arch Biochem Biophys. 2018. PMID: 30077544 Review.
Cited by
-
Intestinal basolateral lipid substrate transport is linked to chylomicron secretion and is regulated by apoC-III.J Lipid Res. 2019 Sep;60(9):1503-1515. doi: 10.1194/jlr.M092460. Epub 2019 May 31. J Lipid Res. 2019. PMID: 31152000 Free PMC article.
-
Regulation of intestinal lipid metabolism: current concepts and relevance to disease.Nat Rev Gastroenterol Hepatol. 2020 Mar;17(3):169-183. doi: 10.1038/s41575-019-0250-7. Epub 2020 Feb 3. Nat Rev Gastroenterol Hepatol. 2020. PMID: 32015520 Review.
-
Effect of the First Feeding on Enterocytes of Newborn Rats.Int J Mol Sci. 2022 Nov 16;23(22):14179. doi: 10.3390/ijms232214179. Int J Mol Sci. 2022. PMID: 36430658 Free PMC article.
-
Emerging Role of Lymphatics in the Regulation of Intestinal Lipid Mobilization.Front Physiol. 2020 Jan 29;10:1604. doi: 10.3389/fphys.2019.01604. eCollection 2019. Front Physiol. 2020. PMID: 32063861 Free PMC article. Review.
-
A novel protein encoded by porcine circANKRD17 activates the PPAR pathway to regulate intramuscular fat metabolism.J Anim Sci Biotechnol. 2025 Feb 5;16(1):19. doi: 10.1186/s40104-025-01153-5. J Anim Sci Biotechnol. 2025. PMID: 39905551 Free PMC article.
References
-
- Nikolopoulou A, Kadoglou NP. Obesity and metabolic syndrome as related to cardiovascular disease. Expert Rev Cardiovasc Ther. 2012;10:933–939. - PubMed
-
- Cases S, Smith SJ, Zheng YW, Myers HM, Lear SR, Sande E, Novak S, Collins C, Welch CB, Lusis AJ, Erickson SK, Farese RV., Jr Identification of a gene encoding an acyl CoA:diacylglycerol acyltransferase, a key enzyme in triacylglycerol synthesis. Proceedings of the National Academy of Sciences of the United States of America. 1998;95:13018–13023. - PMC - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases

