Exo-erythrocytic development of avian malaria and related haemosporidian parasites
- PMID: 28253926
- PMCID: PMC5335495
- DOI: 10.1186/s12936-017-1746-7
Exo-erythrocytic development of avian malaria and related haemosporidian parasites
Abstract
Background: Avian malaria parasites (Plasmodium spp.) and related haemosporidians (Haemosporida) are responsible for diseases which can be severe and even lethal in avian hosts. These parasites cause not only blood pathology, but also damage various organs due to extensive exo-erythrocytic development all over the body, which is not the case during Plasmodium infections in mammals. However, exo-erythrocytic development (tissue merogony or schizogony) remains the most poorly investigated part of life cycle in all groups of wildlife haemosporidian parasites. In spite of remarkable progress in studies of genetic diversity, ecology and evolutionary biology of avian haemosporidians during the past 20 years, there is not much progress in understanding patterns of exo-erythrocytic development in these parasites. The purpose of this review is to overview the main information on exo-erythrocytic development of avian Plasmodium species and related haemosporidian parasites as a baseline for assisting academic and veterinary medicine researchers in morphological identification of these parasites using tissue stages, and to define future research priorities in this field of avian malariology.
Methods: The data were considered from peer-reviewed articles and histological material that was accessed in zoological collections in museums of Australia, Europe and the USA. Articles describing tissue stages of avian haemosporidians were included from 1908 to the present. Histological preparations of various organs infected with the exo-erythrocytic stages of different haemosporidian parasites were examined.
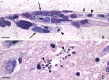
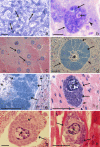
Results: In all, 229 published articles were included in this review. Exo-erythrocytic stages of avian Plasmodium, Fallisia, Haemoproteus, Leucocytozoon, and Akiba species were analysed, compared and illustrated. Morphological characters of tissue stages that can be used for diagnostic purposes were specified.
Conclusion: Recent molecular studies combined with histological research show that avian haemosporidians are more virulent than formerly believed. The exo-erythrocytic stages can cause severe disease, especially in non-adapted avian hosts, suggesting the existence of a group of underestimated malignant infections. The development of a given haemosporidian strain can be markedly different in different avian hosts, resulting in significantly different virulence. A methodology combining the traditional histology techniques with molecular diagnostic tools is essential to speed research in this field of avian malariology.
Keywords: Akiba; Avian malaria; Exo-erythrocytic development; Fallisia; Haemoproteus; Leucocytozoon; Pathology; Plasmodium.
Figures



References
-
- Garnham PCC. Malaria parasites and other Haemosporidia. Oxford: Black-well; 1966.
-
- Seed TM, Manwell RD. Plasmodia of birds. In: Kreier JP, editor. Parasitic Protozoa Vol III. Gregarines, Haemogregarines, Coccidia, Plasmodia, and Haemoproteids. New York: Academic Press; 1977. pp. 311–357.
-
- Valkiūnas G. Avian malaria parasites and other Haemosporidia. Boca Raton: CRC; 2005.
-
- Atkinson CT, Thomas NJ, Hunter DB. Parasitic diseases of wild birds. Oxford: Wiley-Blackwell; 2008.
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

