Photodynamic Detection of Peritoneal Metastases Using 5-Aminolevulinic Acid (ALA)
- PMID: 28257041
- PMCID: PMC5366818
- DOI: 10.3390/cancers9030023
Photodynamic Detection of Peritoneal Metastases Using 5-Aminolevulinic Acid (ALA)
Abstract
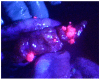
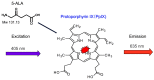
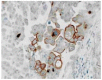
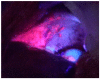
In the past, peritoneal metastasis (PM) was considered as a terminal stage of cancer. From the early 1990s, however, a new comprehensive treatment consisting of cytoreductive surgery and perioperative chemotherapy has been established to improve long-term survival for selected patients with PM. Among prognostic indicators after the treatment, completeness of cytoreduction is the most independent predictors of survival. However, peritoneal recurrence is a main cause of recurrence, even after complete cytoreduction. As a cause of peritoneal recurrence, small PM may be overlooked at the time of cytoreductive surgery (CRS), therefore, development of a new method to detect small PM is desired. Recently, photodynamic diagnosis (PDD) was developed for detection of PM. The objectives of this review were to evaluate whether PDD using 5-aminolevulinic acid (ALA) could improve detection of small PM.
Keywords: ABCG2; PEPT1; aminolevulinic acid; ferrochelatase; peritoneal surface malignancies; photodynamic diagnosis (PDD).
Conflict of interest statement
The authors declare no conflict of interest.
Figures


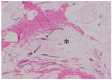


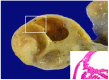
References
-
- Verwaal V.J., Bruin S., Boot H., van Slooten G., van Tinteren H. 8-year follow-up of randomized trial: Cytoreductionand hyperthermic intraperitoneal chemotherapy versus systemic chemotherapy in patients with peritoneal carcinomatosis of colorectal cancer. Ann. Surg. Oncol. 2008;15:2426–2432. doi: 10.1245/s10434-008-9966-2. - DOI - PubMed
-
- Yang X.J., Huang C.Q., Suo T., Mei L.J., Yang G.L., Cheng F.L., Zhou Y.F., Xiong B., Yonemura Y., Li Y. Cytoreductive surgery and hyperthermic intraperitoneal chemotherapy improves survival of patients with peritoneal carcinomatosis from gastric cancer: Final results of a phase III randomized clinical trial. Ann. Surg. Oncol. 2011;18:1575–1581. doi: 10.1245/s10434-011-1631-5. - DOI - PMC - PubMed
Publication types
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous

