Seeing red; the development of pON.mCherry, a broad-host range constitutive expression plasmid for Gram-negative bacteria
- PMID: 28257493
- PMCID: PMC5336243
- DOI: 10.1371/journal.pone.0173116
Seeing red; the development of pON.mCherry, a broad-host range constitutive expression plasmid for Gram-negative bacteria
Abstract
The development of plasmid-mediated gene expression control in bacteria revolutionized the field of bacteriology. Many of these expression control systems rely on the addition of small molecules, generally metabolites or non-metabolized analogs thereof, to the growth medium to induce expression of the genes of interest. The paradigmatic example of an expression control system is the lac system from Escherichia coli, which typically relies on the Ptac promoter and the Lac repressor, LacI. In many cases, however, constitutive gene expression is desired, and other experimental approaches require the coordinated control of multiple genes. While multiple systems have been developed for use in E. coli and its close relatives, the utility and/or functionality of these tools does not always translate to other species. For example, for the Gram-negative pathogen, Legionella pneumophila, a causative agent of Legionnaires' Disease, the aforementioned Ptac system represents the only well-established expression control system. In order to enhance the tools available to study bacterial gene expression in L. pneumophila, we developed a plasmid, pON.mCherry, which confers constitutive gene expression from a mutagenized LacI binding site. We demonstrate that pON.mCherry neither interferes with other plasmids harboring an intact LacI-Ptac expression system nor alters the growth of Legionella species during intracellular growth. Furthermore, the broad-host range plasmid backbone of pON.mCherry allows constitutive gene expression in a wide variety of Gram-negative bacterial species, making pON.mCherry a useful tool for the greater research community.
Conflict of interest statement
Figures
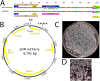
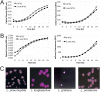
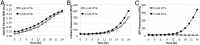
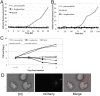
References
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials
Miscellaneous

