Chloroquine improves the response to ischemic muscle injury and increases HMGB1 after arterial ligation
- PMID: 28259568
- PMCID: PMC5581296
- DOI: 10.1016/j.jvs.2017.01.021
Chloroquine improves the response to ischemic muscle injury and increases HMGB1 after arterial ligation
Abstract
Objective: We have previously shown that exogenous administration of the nuclear protein high mobility group box 1 (HMGB1) improves angiogenesis after tissue ischemia. Antagonizing HMGB1 prolongs muscle necrosis and deters regeneration. In this study, we evaluated HMGB1 expression in peripheral arterial disease (PAD) and the mechanisms that promote its release in a murine model of hindlimb ischemia. Specifically, we investigated how chloroquine (CQ), a commonly employed disease-modifying antirheumatic drug, promotes HMGB1 release from muscle. We hypothesized that CQ could increase HMGB1 locally and systemically, allowing it to mediate recovery from ischemic injury.
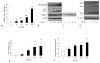
Methods: Muscle biopsies were performed on patients undergoing lower extremity surgery for non-PAD-related disease as well as for claudication and critical limb ischemia. Clinical symptoms and ankle-brachial indices were recorded for each patient. HMGB1 was detected in muscle sections using immunohistochemical staining. Unilateral femoral artery ligation was performed on both wild-type and inducible HMGB1 knockout mice. Wild-type mice were administered intraperitoneal CQ 2 weeks before and after femoral artery ligation. Laser Doppler perfusion imaging was used to determine perfusion recovery. Serum and tissue levels of HMGB1 were measured at designated time points. In vitro, cultured C2C12 myoblasts were treated with increasing doses of CQ. HMGB1, autophagosome formation, p62/SQSTM1 accumulation, caspase-1 expression and activity, and lactate dehydrogenase levels were measured in supernatants and cell lysates.
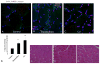
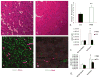
Results: Nuclear expression of HMGB1 was prominent in patients with claudication and critical limb ischemia (P < .05) compared with controls. CQ-treated mice had elevated serum HMGB1 and diffuse HMGB1 staining in muscle (P < .01). In wild-type mice, CQ treatment resulted in higher laser Doppler perfusion imaging ratios in the ischemic limb at 7 days (P < .03) and less fat replacement after 2 weeks (P < .03). In cultured myoblasts, CQ induced autophagosome accumulation, inhibited p62/SQSTM-1 degradation, and activated caspase-1.
Conclusions: HMGB1 is prominently expressed in PAD muscle but mostly confined to the nucleus. Our in vivo data suggest that HMGB1 mobilization into the sarcoplasm and serum can be increased with CQ, possibly through caspase-1-mediated pathways. Whereas HMGB1 can be released by many cell types, these studies suggest that the muscle may be an important additional source that is relevant in PAD.
Copyright © 2017 Society for Vascular Surgery. All rights reserved.
Figures




Similar articles
-
Caspase1/11 signaling affects muscle regeneration and recovery following ischemia, and can be modulated by chloroquine.Mol Med. 2020 Jul 8;26(1):69. doi: 10.1186/s10020-020-00190-2. Mol Med. 2020. PMID: 32641037 Free PMC article.
-
HMGB1 and TLR4 mediate skeletal muscle recovery in a murine model of hindlimb ischemia.J Vasc Surg. 2013 Aug;58(2):460-9. doi: 10.1016/j.jvs.2012.11.071. Epub 2013 Feb 12. J Vasc Surg. 2013. PMID: 23414695 Free PMC article.
-
High mobility group box 1 promotes endothelial cell angiogenic behavior in vitro and improves muscle perfusion in vivo in response to ischemic injury.J Vasc Surg. 2012 Jan;55(1):180-91. doi: 10.1016/j.jvs.2011.07.072. Epub 2011 Sep 23. J Vasc Surg. 2012. PMID: 21944908 Free PMC article.
-
Skeletal Muscle Pathology in Peripheral Artery Disease: A Brief Review.Arterioscler Thromb Vasc Biol. 2020 Nov;40(11):2577-2585. doi: 10.1161/ATVBAHA.120.313831. Epub 2020 Sep 17. Arterioscler Thromb Vasc Biol. 2020. PMID: 32938218 Free PMC article. Review.
-
Systematic Interrogation of Angiogenesis in the Ischemic Mouse Hind Limb: Vulnerabilities and Quality Assurance.Arterioscler Thromb Vasc Biol. 2020 Oct;40(10):2454-2467. doi: 10.1161/ATVBAHA.120.315028. Epub 2020 Aug 13. Arterioscler Thromb Vasc Biol. 2020. PMID: 32787524 Free PMC article.
Cited by
-
Fibrosis Distinguishes Critical Limb Ischemia Patients from Claudicants in a Transcriptomic and Histologic Analysis.J Clin Med. 2020 Dec 8;9(12):3974. doi: 10.3390/jcm9123974. J Clin Med. 2020. PMID: 33302519 Free PMC article.
-
Inflammatory Caspase Activity Mediates HMGB1 Release and Differentiation in Myoblasts Affected by Peripheral Arterial Disease.Cells. 2022 Mar 30;11(7):1163. doi: 10.3390/cells11071163. Cells. 2022. PMID: 35406727 Free PMC article.
-
Chloroquine commonly induces hormetic dose responses.Sci Total Environ. 2021 Feb 10;755(Pt 1):142436. doi: 10.1016/j.scitotenv.2020.142436. Epub 2020 Sep 25. Sci Total Environ. 2021. PMID: 33017762 Free PMC article.
-
High Mobility Group Box-1 and Diabetes Mellitus Complications: State of the Art and Future Perspectives.Int J Mol Sci. 2019 Dec 11;20(24):6258. doi: 10.3390/ijms20246258. Int J Mol Sci. 2019. PMID: 31835864 Free PMC article. Review.
-
Caspase1/11 signaling affects muscle regeneration and recovery following ischemia, and can be modulated by chloroquine.Mol Med. 2020 Jul 8;26(1):69. doi: 10.1186/s10020-020-00190-2. Mol Med. 2020. PMID: 32641037 Free PMC article.
References
-
- Selvin E, Erlinger TP. Prevalence of and risk factors for peripheral arterial disease in the United States: results from the National Health and Nutrition Examination Survey, 1999–2000. Circulation. 2004;110:738–43. - PubMed
-
- Nehler MR, Duval S, Diao L, Annex BH, Hiatt WR, Rogers K, Zakharyan A, Hirsch AT. Epidemiology of peripheral arterial disease and critical limb ischemia in an insured national population. Journal of Vascular Surgery. 2014;60:686–695. e2. - PubMed
-
- Rajagopalan S, Mohler ER, Lederman RJ, Mendelsohn FO, Saucedo JF, Goldman CK, Blebea J, Macko J, Kessler PD, Rasmussen HS, Annex BH. Regional Angiogenesis With Vascular Endothelial Growth Factor in Peripheral Arterial Disease: A Phase II Randomized, Double-Blind, Controlled Study of Adenoviral Delivery of Vascular Endothelial Growth Factor 121 in Patients With Disabling Intermittent Claudication. Circulation. 2003;108:1933–1938. - PubMed
-
- Masaki I, Yonemitsu Y, Yamashita A, Sata S, Tanii M, Komori K, Nakagawa K, Hou X, Nagai Y, Hasegawa M, Sugimachi K, Sueishi K. Angiogenic gene therapy for experimental critical limb ischemia: acceleration of limb loss by overexpression of vascular endothelial growth factor 165 but not of fibroblast growth factor-2. Circ Res. 2002;90:966–73. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases

