Are child-centric aspects in newborn and child health systematic review and meta-analysis protocols and reports adequately reported?-two systematic reviews
- PMID: 28260528
- PMCID: PMC5338085
- DOI: 10.1186/s13643-017-0423-9
Are child-centric aspects in newborn and child health systematic review and meta-analysis protocols and reports adequately reported?-two systematic reviews
Abstract
Background: Evidence suggests that newborn and child health systematic reviews and meta-analyses exhibit poor quality in reporting. The "Preferred Reporting Items in Systematic Review and Meta-Analysis" (PRISMA) and PRISMA-Protocols (PRISMA-P) checklists have been developed to improve the reporting of systematic review results and protocols, respectively. We aimed to evaluate the clarity and transparency in reporting of child-centric items in child health systematic reviews (SRs) and SR protocols and to identify areas where reporting could be strengthened.
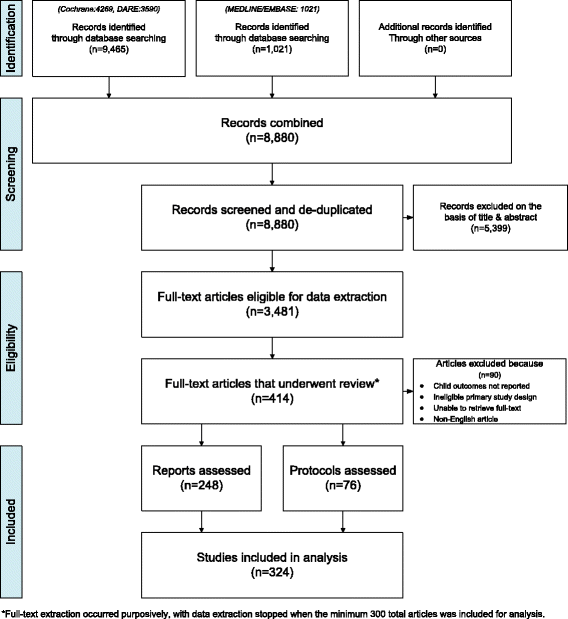
Methods: Two preliminary lists of potential child-centric reporting items were used to examine current reporting. The Cochrane, DARE, MEDLINE, and EMBASE libraries were searched from 2010 to 2014 for systematic reviews that included children. Each report and protocol that met the inclusion criteria had their quality of reporting assessed by their reporting of child-centric items. Quality of reporting was assessed per whether one third, one to two thirds, or more than two thirds of papers complied with potential child-centric potential modifications/extensions to PRISMA and were analyzed by the following: (i) paper type (i.e., report vs. protocol), (ii) publication type (i.e., Cochrane vs. non-Cochrane), and (iii) population type (i.e., child-only vs. mixed populations vs. family/maternal).
Results: Of the 414 eligible articles, 248 reports and 76 protocols were included. In 21 of 24 potential SR reporting items and 13 of 14 potential SR protocol reporting items, less than two thirds of papers met the child-centric reporting item requirements. Mixed population studies displayed significantly poorer reporting in comparison to child-only and family/maternal intervention studies for 11 potential SR reporting items (p < 0.05) and five potential SR protocol items (p < 0.05). When comparing non-Cochrane to Cochrane reports and protocols, five items in both lists were found to perform significantly poorer in non-Cochrane reports (p < 0.05). Significant differences in reporting quality were found in three of 14 items shared between the potential SR reporting items and potential SR protocol reporting items (p < 0.05).
Conclusions: Newborn and child health systematic reviews and meta-analyses exhibit incomplete reporting, thereby hindering prudent decision-making by healthcare providers and policy makers. These results provide a rationale for the implementation of child-centric extensions and modifications to current PRISMA and PRISMA-P, such as to improve reporting in this population.
Keywords: Child health; PRISMA; PRISMA-P; Protocol; Reporting guidelines; Systematic review.
Figures
References
-
- OCEBM Levels of evidence working group. The Oxford 2011 Levels of Evidence. Oxford Centre for Evidence-Based Medicine; 2011.
-
- Hutton B, Salanti G, Caldwell DM, Chaimani A, Schmid CH, Cameron C, Ioannidis JPA, Straus S, Thorlund K, Jansen JP, Mulrow C, Catalá-López F, Gøtzsche PC, Dickersin K, Boutron I, Altman DG, Moher D. The PRISMA extension statement for reporting of systematic reviews incorporating network meta-analyses of health care interventions: checklist and explanations. Ann Intern Med. 2015;162:777–784. doi: 10.7326/M14-2385. - DOI - PubMed
MeSH terms
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials
Miscellaneous