Plant immune and growth receptors share common signalling components but localise to distinct plasma membrane nanodomains
- PMID: 28262094
- PMCID: PMC5383397
- DOI: 10.7554/eLife.25114
Plant immune and growth receptors share common signalling components but localise to distinct plasma membrane nanodomains
Abstract
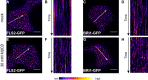
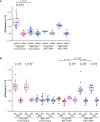
Cell surface receptors govern a multitude of signalling pathways in multicellular organisms. In plants, prominent examples are the receptor kinases FLS2 and BRI1, which activate immunity and steroid-mediated growth, respectively. Intriguingly, despite inducing distinct signalling outputs, both receptors employ common downstream signalling components, which exist in plasma membrane (PM)-localised protein complexes. An important question is thus how these receptor complexes maintain signalling specificity. Live-cell imaging revealed that FLS2 and BRI1 form PM nanoclusters. Using single-particle tracking we could discriminate both cluster populations and we observed spatiotemporal separation between immune and growth signalling platforms. This finding was confirmed by visualising FLS2 and BRI1 within distinct PM nanodomains marked by specific remorin proteins and differential co-localisation with the cytoskeleton. Our results thus suggest that signalling specificity between these pathways may be explained by the spatial separation of FLS2 and BRI1 with their associated signalling components within dedicated PM nanodomains.
Keywords: A. thaliana; cell biology; growth; immunity; microdomain; plant biology; plasma membrane; receptor kinase; signaling.
Conflict of interest statement
The authors declare that no competing interests exist.
Figures

















Similar articles
-
Myosin XI-mediated BIK1 recruitment to nanodomains facilitates FLS2-BIK1 complex formation during innate immunity in Arabidopsis.Proc Natl Acad Sci U S A. 2024 Jun 18;121(25):e2312415121. doi: 10.1073/pnas.2312415121. Epub 2024 Jun 14. Proc Natl Acad Sci U S A. 2024. PMID: 38875149 Free PMC article.
-
Visualization of BRI1 and SERK3/BAK1 Nanoclusters in Arabidopsis Roots.PLoS One. 2017 Jan 23;12(1):e0169905. doi: 10.1371/journal.pone.0169905. eCollection 2017. PLoS One. 2017. PMID: 28114413 Free PMC article.
-
Super-Resolution Imaging of Plant Receptor-Like Kinases Uncovers Their Colocalization and Coordination with Nanometer Resolution.Membranes (Basel). 2023 Jan 21;13(2):142. doi: 10.3390/membranes13020142. Membranes (Basel). 2023. PMID: 36837645 Free PMC article.
-
Dynamic complexity: plant receptor complexes at the plasma membrane.Curr Opin Plant Biol. 2017 Dec;40:15-21. doi: 10.1016/j.pbi.2017.06.016. Epub 2017 Jul 14. Curr Opin Plant Biol. 2017. PMID: 28715768 Review.
-
Computational modelling of the BRI1 receptor system.Plant Cell Environ. 2013 Sep;36(9):1728-37. doi: 10.1111/pce.12077. Epub 2013 Mar 12. Plant Cell Environ. 2013. PMID: 23421559 Review.
Cited by
-
A molecular roadmap to the plant immune system.J Biol Chem. 2020 Oct 30;295(44):14916-14935. doi: 10.1074/jbc.REV120.010852. Epub 2020 Aug 17. J Biol Chem. 2020. PMID: 32816993 Free PMC article. Review.
-
Battlefield Cytoskeleton: Turning the Tide on Plant Immunity.Mol Plant Microbe Interact. 2019 Jan;32(1):25-34. doi: 10.1094/MPMI-07-18-0195-FI. Epub 2018 Oct 24. Mol Plant Microbe Interact. 2019. PMID: 30355064 Free PMC article. Review.
-
Phosphorylation-Mediated Dynamics of Nitrate Transceptor NRT1.1 Regulate Auxin Flux and Nitrate Signaling in Lateral Root Growth.Plant Physiol. 2019 Oct;181(2):480-498. doi: 10.1104/pp.19.00346. Epub 2019 Aug 20. Plant Physiol. 2019. PMID: 31431511 Free PMC article.
-
Tomato Dynamin Related Protein 2A Associates With LeEIX2 and Enhances PRR Mediated Defense by Modulating Receptor Trafficking.Front Plant Sci. 2019 Jul 19;10:936. doi: 10.3389/fpls.2019.00936. eCollection 2019. Front Plant Sci. 2019. PMID: 31379912 Free PMC article.
-
Regulation of Arabidopsis brassinosteroid receptor BRI1 endocytosis and degradation by plant U-box PUB12/PUB13-mediated ubiquitination.Proc Natl Acad Sci U S A. 2018 Feb 20;115(8):E1906-E1915. doi: 10.1073/pnas.1712251115. Epub 2018 Feb 5. Proc Natl Acad Sci U S A. 2018. PMID: 29432171 Free PMC article.
References
-
- Albrecht C, Boutrot F, Segonzac C, Schwessinger B, Gimenez-Ibanez S, Chinchilla D, Rathjen JP, de Vries SC, Zipfel C. Brassinosteroids inhibit pathogen-associated molecular pattern-triggered immune signaling independent of the receptor kinase BAK1. PNAS. 2012;109:303–308. doi: 10.1073/pnas.1109921108. - DOI - PMC - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources

