Molecular basis of ancestral vertebrate electroreception
- PMID: 28264196
- PMCID: PMC5354974
- DOI: 10.1038/nature21401
Molecular basis of ancestral vertebrate electroreception
Abstract
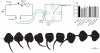
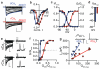
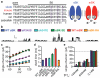
Elasmobranch fishes, including sharks, rays, and skates, use specialized electrosensory organs called ampullae of Lorenzini to detect extremely small changes in environmental electric fields. Electrosensory cells within these ampullae can discriminate and respond to minute changes in environmental voltage gradients through an unknown mechanism. Here we show that the voltage-gated calcium channel CaV1.3 and the big conductance calcium-activated potassium (BK) channel are preferentially expressed by electrosensory cells in little skate (Leucoraja erinacea) and functionally couple to mediate electrosensory cell membrane voltage oscillations, which are important for the detection of specific, weak electrical signals. Both channels exhibit unique properties compared with their mammalian orthologues that support electrosensory functions: structural adaptations in CaV1.3 mediate a low-voltage threshold for activation, and alterations in BK support specifically tuned voltage oscillations. These findings reveal a molecular basis of electroreception and demonstrate how discrete evolutionary changes in ion channel structure facilitate sensory adaptation.
Figures


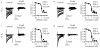










Comment in
-
Sensory Systems: Electrifying News from the Ocean.Curr Biol. 2017 Dec 18;27(24):R1327-R1329. doi: 10.1016/j.cub.2017.11.015. Curr Biol. 2017. PMID: 29257970
References
-
- Kalmijn AJ. The electric sense of sharks and rays. The Journal of Experimental Biology. 1971;55:371–383. - PubMed
-
- Kalmijn AJ. Electric and magnetic field detection in elasmobranch fishes. Science. 1982;218:916–918. - PubMed
-
- Lissmann HW, Machin KE. The mechanism of object location in Gymnarchus niloticus and similar fish. Journal of Experimental Biology. 1958;35:451–486.
-
- Munz H, Claas B, Fritzsch B. Electroreceptive and mechanoreceptive units in the lateral line of the axolotl Ambystoma mexicanum. J Comp Physiol. 1984;154:33–44.
-
- Scheich H, Langner G, Tidemann C, Coles RB, Guppy A. Electroreception and electrolocation in platypus. Nature. 1986;319:401–402. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials

