The Pharmaceutical Industry in 2016. An Analysis of FDA Drug Approvals from a Perspective of the Molecule Type
- PMID: 28264468
- PMCID: PMC6155368
- DOI: 10.3390/molecules22030368
The Pharmaceutical Industry in 2016. An Analysis of FDA Drug Approvals from a Perspective of the Molecule Type
Abstract
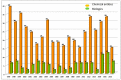
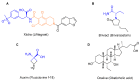
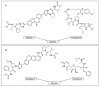
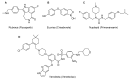
This is an analysis from a chemical point of view of the 22 drugs accepted by the FDA during 2016. The different drugs from the 2016 "harvest" have been classified according to their chemical structure: antibodies; TIDES (oligonucleotides and peptides); amino acids and natural products; drug combination; and small molecules.
Keywords: API; biologics; chemical entities; drug discovery; peptide; small molecules.
Conflict of interest statement
The authors declare no conflict of interest.
Figures


References
-
- Joint Research Centre European Commission . The 2016 EU Industrial R&D Investment Scoreboard. Publications Office of the EU; London, UK: 2016.
-
- U.S. Food and Drug Administration (FDA) [(accessed on 5 February 2017)]; Available online: http://www.accessdata.fda.gov/scripts/cder/daf/
-
- Jarvis L.M. The Year in New Drugs. Chemical & Engineering News; Washington, DC, USA: 2017. pp. 28–32.
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

