Deletion of the Fractalkine Receptor, CX3CR1, Improves Endogenous Repair, Axon Sprouting, and Synaptogenesis after Spinal Cord Injury in Mice
- PMID: 28264978
- PMCID: PMC5373135
- DOI: 10.1523/JNEUROSCI.2841-16.2017
Deletion of the Fractalkine Receptor, CX3CR1, Improves Endogenous Repair, Axon Sprouting, and Synaptogenesis after Spinal Cord Injury in Mice
Abstract
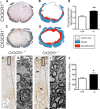
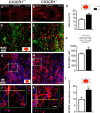
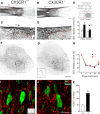
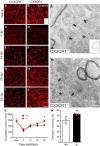
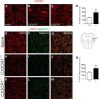
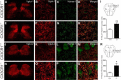
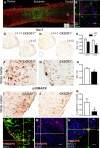
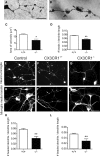
Impaired signaling via CX3CR1, the fractalkine receptor, promotes recovery after traumatic spinal contusion injury in mice, a benefit achieved in part by reducing macrophage-mediated injury at the lesion epicenter. Here, we tested the hypothesis that CX3CR1-dependent changes in microglia and macrophage functions also will enhance neuroplasticity, at and several segments below the injury epicenter. New data show that in the presence of inflammatory stimuli, CX3CR1-deficient (CX3CR1-/-) microglia and macrophages adopt a reparative phenotype and increase expression of genes that encode neurotrophic and gliogenic proteins. At the lesion epicenter (mid-thoracic spinal cord), the microenvironment created by CX3CR1-/- microglia/macrophages enhances NG2 cell responses, axon sparing, and sprouting of serotonergic axons. In lumbar spinal cord, inflammatory signaling is reduced in CX3CR1-/- microglia. This is associated with reduced dendritic pathology and improved axonal and synaptic plasticity on ventral horn motor neurons. Together, these data indicate that CX3CR1, a microglia-specific chemokine receptor, is a novel therapeutic target for enhancing neuroplasticity and recovery after SCI. Interventions that specifically target CX3CR1 could reduce the adverse effects of inflammation and augment activity-dependent plasticity and restoration of function. Indeed, limiting CX3CR1-dependent signaling could improve rehabilitation and spinal learning.SIGNIFICANCE STATEMENT Published data show that genetic deletion of CX3CR1, a microglia-specific chemokine receptor, promotes recovery after traumatic spinal cord injury in mice, a benefit achieved in part by reducing macrophage-mediated injury at the lesion epicenter. Data in the current manuscript indicate that CX3CR1 deletion changes microglia and macrophage function, creating a tissue microenvironment that enhances endogenous repair and indices of neuroplasticity, at and several segments below the injury epicenter. Interventions that specifically target CX3CR1 might be used in the future to reduce the adverse effects of intraspinal inflammation and augment activity-dependent plasticity (e.g., rehabilitation) and restoration of function.
Keywords: CX3CR1; inflammation; macrophages; microglia; plasticity; spinal cord injury.
Copyright © 2017 the authors 0270-6474/17/373568-20$15.00/0.
Figures


Similar articles
-
miR-155 Deletion in Mice Overcomes Neuron-Intrinsic and Neuron-Extrinsic Barriers to Spinal Cord Repair.J Neurosci. 2016 Aug 10;36(32):8516-32. doi: 10.1523/JNEUROSCI.0735-16.2016. J Neurosci. 2016. PMID: 27511021 Free PMC article.
-
Deficient CX3CR1 signaling promotes recovery after mouse spinal cord injury by limiting the recruitment and activation of Ly6Clo/iNOS+ macrophages.J Neurosci. 2011 Jul 6;31(27):9910-22. doi: 10.1523/JNEUROSCI.2114-11.2011. J Neurosci. 2011. PMID: 21734283 Free PMC article.
-
High-resolution intravital imaging reveals that blood-derived macrophages but not resident microglia facilitate secondary axonal dieback in traumatic spinal cord injury.Exp Neurol. 2014 Apr;254:109-20. doi: 10.1016/j.expneurol.2014.01.013. Epub 2014 Jan 24. Exp Neurol. 2014. PMID: 24468477 Free PMC article.
-
Analysis of the Role of CX3CL1 (Fractalkine) and Its Receptor CX3CR1 in Traumatic Brain and Spinal Cord Injury: Insight into Recent Advances in Actions of Neurochemokine Agents.Mol Neurobiol. 2017 Apr;54(3):2167-2188. doi: 10.1007/s12035-016-9787-4. Epub 2016 Mar 1. Mol Neurobiol. 2017. PMID: 26927660 Free PMC article. Review.
-
Macrophage activation and its role in repair and pathology after spinal cord injury.Brain Res. 2015 Sep 4;1619:1-11. doi: 10.1016/j.brainres.2014.12.045. Epub 2015 Jan 8. Brain Res. 2015. PMID: 25578260 Review.
Cited by
-
Resident immune responses to spinal cord injury: role of astrocytes and microglia.Neural Regen Res. 2024 Aug 1;19(8):1678-1685. doi: 10.4103/1673-5374.389630. Epub 2023 Dec 11. Neural Regen Res. 2024. PMID: 38103231 Free PMC article.
-
Mesenchymal stem cell-derived exosomes containing miR-145-5p reduce inflammation in spinal cord injury by regulating the TLR4/NF-κB signaling pathway.Cell Cycle. 2021 May;20(10):993-1009. doi: 10.1080/15384101.2021.1919825. Epub 2021 May 4. Cell Cycle. 2021. PMID: 33945431 Free PMC article.
-
Eliciting inflammation enables successful rehabilitative training in chronic spinal cord injury.Brain. 2018 Jul 1;141(7):1946-1962. doi: 10.1093/brain/awy128. Brain. 2018. PMID: 29860396 Free PMC article.
-
Conditional RAC1 knockout in motor neurons restores H-reflex rate-dependent depression after spinal cord injury.Sci Rep. 2021 Apr 9;11(1):7838. doi: 10.1038/s41598-021-87476-5. Sci Rep. 2021. PMID: 33837249 Free PMC article.
-
CSF IL-8 Associated with Response to Gene Therapy in a Case Series of Spinal Muscular Atrophy.Neurotherapeutics. 2023 Jan;20(1):245-253. doi: 10.1007/s13311-022-01305-9. Epub 2022 Oct 26. Neurotherapeutics. 2023. PMID: 36289175 Free PMC article.
References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases
Research Materials
