Screening for Bowel Cancer: Increasing Participation via Personal Invitation
- PMID: 28266301
- PMCID: PMC5341111
- DOI: 10.3238/arztebl.2017.0087
Screening for Bowel Cancer: Increasing Participation via Personal Invitation
Abstract
Background: Participation rates in bowel cancer screening programs in Germany continue to be low. In a model project, a logistically simple procedure for inviting patients to participate was tested as a means of increasing participation.
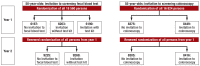
Methods: A randomized trial was performed involving persons residing in the German federal state of Saarland who had either their 50th or their 55th birthday in the year beginning on 1 April 2012 (18 560 and 16 824 persons, respectively). The 50-year-olds received a written invitation to undergo a test for blood in the stool, either with or without a stool test attached, or else no invitation at all. The 55-year-olds received either an invitation to undergo colonoscopy or no invitation. Participation rates within one year were determined from billing data of the Saarland Association of Statutory Health. Insurance Physicians. The trial was registered in the German Registry of Clinical Trials, no. DRKS00006098.
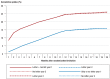
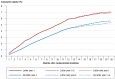
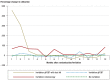
Results: A written invitation to undergo testing of the stool for blood, together with an accompanying test, increased the participation rate within one year by 62% (from 15% to 25%, p <0.001, especially among men (+158% vs. +39% for women). The participation rate was higher in general among women than among men (33% vs. 17%). On the other hand, a written invitation with no accompanying test did not increase the participation rate. A written invitation to undergo colonoscopic screening increased the participation rate within one year by 32% (5.9% vs 4.4%, p <0.001).
Conclusion: Targeted invitations can markedly increase participation rates in cancer screening. Written invitations to undergo stool testing for blood should be accompanied by an actual test. Further trials should also include information about the number of adenomas and carcinomas detected by screening.
Figures
Comment in
-
Missing Information.Dtsch Arztebl Int. 2017 Jun 23;114(25):426. doi: 10.3238/arztebl.2017.0426a. Dtsch Arztebl Int. 2017. PMID: 28683854 Free PMC article. No abstract available.
-
Preventing Bowel Cancer Through Hormones.Dtsch Arztebl Int. 2017 Jun 23;114(25):426-427. doi: 10.3238/arztebl.2017.0426b. Dtsch Arztebl Int. 2017. PMID: 28683855 Free PMC article. No abstract available.
References
-
- Robert Koch-Institut, Gesellschaft der epidemiologischen Krebsregister in Deutschland e. V. (eds.) Häufigkeiten und Trends. 10. Berlin: 2015. Krebs in Deutschland 2011/2012.
-
- Hewitson P, Glasziou P, Watson E, Towler B, Irwig L. Cochrane systematic review of colorectal cancer screening using the fecal occult blood test (Hemoccult): an update. Am J Gastroenterol. 2008;103:1541–1549. - PubMed
-
- Robertson DJ, Kaminski MF, Bretthauer M. Effectiveness, training and quality assurance of colonoscopy screening for colorectal cancer. Gut. 2015;64:982–990. - PubMed
-
- Winawer SJ, Zauber AG, Ho MN, et al. Prevention of colorectal cancer by colonoscopic polypectomy. The National Polyp Study Workgroup. N Engl J Med. 1993;329:1977–1981. - PubMed
-
- Walsh JM, Terdiman JP. Colorectal cancer screening: scientific review. JAMA. 2003;289:1288–1296. - PubMed
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Miscellaneous

