A Reverse Genetics Platform That Spans the Zika Virus Family Tree
- PMID: 28270583
- PMCID: PMC5340872
- DOI: 10.1128/mBio.02014-16
A Reverse Genetics Platform That Spans the Zika Virus Family Tree
Abstract
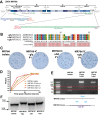
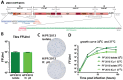
Zika virus (ZIKV), a mosquito-borne flavivirus discovered in 1947, has only recently caused large outbreaks and emerged as a significant human pathogen. In 2015, ZIKV was detected in Brazil, and the resulting epidemic has spread throughout the Western Hemisphere. Severe complications from ZIKV infection include neurological disorders such as Guillain-Barré syndrome in adults and a variety of fetal abnormalities, including microcephaly, blindness, placental insufficiency, and fetal demise. There is an urgent need for tools and reagents to study the pathogenesis of epidemic ZIKV and for testing vaccines and antivirals. Using a reverse genetics platform, we generated six ZIKV infectious clones and derivative viruses representing diverse temporal and geographic origins. These include three versions of MR766, the prototype 1947 strain (with and without a glycosylation site in the envelope protein), and H/PF/2013, a 2013 human isolate from French Polynesia representative of the virus introduced to Brazil. In the course of synthesizing a clone of a circulating Brazilian strain, phylogenetic studies identified two distinct ZIKV clades in Brazil. We reconstructed viable clones of strains SPH2015 and BeH819015, representing ancestral members of each clade. We assessed recombinant virus replication, binding to monoclonal antibodies, and virulence in mice. This panel of molecular clones and recombinant virus isolates will enable targeted studies of viral determinants of pathogenesis, adaptation, and evolution, as well as the rational attenuation of contemporary outbreak strains to facilitate the design of vaccines and therapeutics.IMPORTANCE Viral emergence is a poorly understood process as evidenced by the sudden emergence of Zika virus in Latin America and the Caribbean. Malleable reagents that both predate and span an expanding epidemic are key to understanding the virologic determinants that regulate pathogenesis and transmission. We have generated representative cDNA molecular clones and recombinant viruses that span the known ZIKV family tree, including early Brazilian isolates. Recombinant viruses replicated efficiently in cell culture and were pathogenic in immunodeficient mice, providing a genetic platform for rational vaccine and therapeutic design.
Copyright © 2017 Widman et al.
Figures


References
-
- Duffy MR, Chen TH, Hancock WT, Powers AM, Kool JL, Lanciotti RS, Pretrick M, Marfel M, Holzbauer S, Dubray C, Guillaumot L, Griggs A, Bel M, Lambert AJ, Laven J, Kosoy O, Panella A, Biggerstaff BJ, Fischer M, Hayes EB. 2009. Zika virus outbreak on Yap Island, Federated States of Micronesia. N Engl J Med 360:2536–2543. doi:10.1056/NEJMoa0805715. - DOI - PubMed
-
- Faria NR, Azevedo RDS, Kraemer MU, Souza R, Cunha MS, Hill SC, Theze J, Bonsall MB, Bowden TA, Rissanen I, Rocco IM, Nogueira JS, Maeda AY, Vasami FG, Macedo FL, Suzuki A, Rodrigues SG, Cruz AC, Nunes BT, Medeiros DB, Rodrigues DS, Nunes Queiroz AL, da Silva EV, Henriques DF, Travassos da Rosa ES, de Oliveira CS, Martins LC, Vasconcelos HB, Casseb LM, Simith DDB, Messina JP, Abade L, Lourenco J, Alcantara LCJ, de Lima MM, Giovanetti M, Hay SI, de Oliveira RS, Lemos PDS, de Oliveira LF, de Lima CP, da Silva SP, de Vasconcelos JM, Franco L, Cardoso JF, Vianez-Junior JL, Mir D, Bello G, Delatorre E, Khan K, Creatore M, Coelho GE, de Oliveira WK, Tesh R, Pybus OG, Nunes MR, Vasconcelos PF. 2016. Zika virus in the Americas: early epidemiological and genetic findings. Science 352:345–349. doi:10.1126/science.aaf5036. - DOI - PMC - PubMed
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

