Decerebrate mouse model for studies of the spinal cord circuits
- PMID: 28277546
- PMCID: PMC8495891
- DOI: 10.1038/nprot.2017.001
Decerebrate mouse model for studies of the spinal cord circuits
Abstract
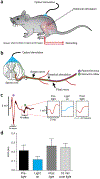
The adult decerebrate mouse model (a mouse with the cerebrum removed) enables the study of sensory-motor integration and motor output from the spinal cord for several hours without compromising these functions with anesthesia. For example, the decerebrate mouse is ideal for examining locomotor behavior using intracellular recording approaches, which would not be possible using current anesthetized preparations. This protocol describes the steps required to achieve a low-blood-loss decerebration in the mouse and approaches for recording signals from spinal cord neurons with a focus on motoneurons. The protocol also describes an example application for the protocol: the evocation of spontaneous and actively driven stepping, including optimization of these behaviors in decerebrate mice. The time taken to prepare the animal and perform a decerebration takes ∼2 h, and the mice are viable for up to 3-8 h, which is ample time to perform most short-term procedures. These protocols can be modified for those interested in cardiovascular or respiratory function in addition to motor function and can be performed by trainees with some previous experience in animal surgery.
Conflict of interest statement
Figures
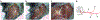

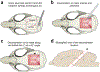
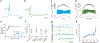


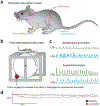
References
-
- Kiehn O Development and functional organization of spinal locomotor circuits. Curr. Opin. Neurobiol 21, 100–109 (2011). - PubMed
-
- Dougherty KJ et al. Locomotor rhythm generation linked to the output of spinal shox2 excitatory interneurons. Neuron 80, 920–933 (2013). - PubMed
-
- Lanuza GM, Gosgnach S, Pierani A, Jessell TM & Goulding M Genetic identification of spinal interneurons that coordinate left-right locomotor activity necessary for walking movements. Neuron 42, 375–386 (2004). - PubMed
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

