Local Activation of p53 in the Tumor Microenvironment Overcomes Immune Suppression and Enhances Antitumor Immunity
- PMID: 28280037
- PMCID: PMC5465961
- DOI: 10.1158/0008-5472.CAN-16-2832
Local Activation of p53 in the Tumor Microenvironment Overcomes Immune Suppression and Enhances Antitumor Immunity
Abstract
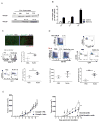
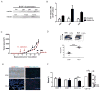
Mutations in tumor suppressor p53 remain a vital mechanism of tumor escape from apoptosis and senescence. Emerging evidence suggests that p53 dysfunction also fuels inflammation and supports tumor immune evasion, thereby serving as an immunological driver of tumorigenesis. Therefore, targeting p53 in the tumor microenvironment (TME) also represents an immunologically desirable strategy for reversing immunosuppression and enhancing antitumor immunity. Using a pharmacological p53 activator nutlin-3a, we show that local p53 activation in TME comprising overt tumor-infiltrating leukocytes (TILeus) induces systemic antitumor immunity and tumor regression, but not in TME with scarce TILeus, such as B16 melanoma. Maneuvers that recruit leukocytes to TME, such as TLR3 ligand in B16 tumors, greatly enhanced nutlin-induced antitumor immunity and tumor control. Mechanistically, nutlin-3a-induced antitumor immunity was contingent on two nonredundant but immunologically synergistic p53-dependent processes: reversal of immunosuppression in the TME and induction of tumor immunogenic cell death, leading to activation and expansion of polyfunctional CD8 CTLs and tumor regression. Our study demonstrates that unlike conventional tumoricidal therapies, which rely on effective p53 targeting in each tumor cell and often associate with systemic toxicity, this immune-based strategy requires only limited local p53 activation to alter the immune landscape of TME and subsequently amplify immune response to systemic antitumor immunity. Hence, targeting the p53 pathway in TME can be exploited to reverse immunosuppression and augment therapeutic benefits beyond tumoricidal effects to harness tumor-specific, durable, and systemic antitumor immunity with minimal toxicity. Cancer Res; 77(9); 2292-305. ©2017 AACR.
©2017 American Association for Cancer Research.
Figures





References
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials
Miscellaneous

