WNK1 is an unexpected autophagy inhibitor
- PMID: 28282258
- PMCID: PMC5446055
- DOI: 10.1080/15548627.2017.1286431
WNK1 is an unexpected autophagy inhibitor
Abstract
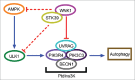
Autophagy is a cellular degradation pathway that is essential to maintain cellular physiology, and deregulation of autophagy leads to multiple diseases in humans. In a recent study, we discovered that the protein kinase WNK1 (WNK lysine deficient protein kinase 1) is an inhibitor of autophagy. The loss of WNK1 increases both basal and starvation-induced autophagy. In addition, the depletion of WNK1 increases the activation of the class III phosphatidylinositol 3-kinase (PtdIns3K) complex, which is required to induce autophagy. Moreover, the loss of WNK1 increases the expression of ULK1 (unc-51 like kinase 1), which is upstream of the PtdIns3K complex. It also increases the pro-autophagic phosphorylation of ULK1 at Ser555 and the activation of AMPK (AMP-activated protein kinase), which is responsible for that phosphorylation. The inhibition of AMPK by compound C decreases the magnitude of autophagy induction following WNK1 loss; however, it does not prevent autophagy induction. We found that the UVRAG (UV radiation resistance associated gene), which is a component of the PtdIns3K, binds to the N-terminal region of WNK1. Moreover, WNK1 partially colocalizes with UVRAG and this colocalization decreases when autophagy is stimulated in cells. The loss of WNK1 also alters the cellular distribution of UVRAG. The depletion of the downstream target of WNK1, OXSR1/OSR1 (oxidative-stress responsive 1) has no effect on autophagy, whereas the depletion of its relative STK39/SPAK (serine/threonine kinase 39) induces autophagy under nutrient-rich and starved conditions.
Keywords: AMPK; OSR1; PI3KC3; SPAK; ULK1; UVRAG; Vps34; with no lysine.
Figures
Comment on
- Punctum to: Gallolu Kankanamalage S., et al., Multistep regulation of autophagy by WNK1. Proc Natl Acad Sci U S A, 2016. 113(50): p. 14342–14347; http://dx.doi.org/ 10.1073/pnas.1617649113.
Similar articles
-
Multistep regulation of autophagy by WNK1.Proc Natl Acad Sci U S A. 2016 Dec 13;113(50):14342-14347. doi: 10.1073/pnas.1617649113. Epub 2016 Nov 28. Proc Natl Acad Sci U S A. 2016. PMID: 27911840 Free PMC article.
-
Suppression of WNK1-SPAK/OSR1 Attenuates Bone Cancer Pain by Regulating NKCC1 and KCC2.J Pain. 2019 Dec;20(12):1416-1428. doi: 10.1016/j.jpain.2019.05.005. Epub 2019 May 11. J Pain. 2019. PMID: 31085334
-
WNK1 and OSR1 regulate the Na+, K+, 2Cl- cotransporter in HeLa cells.Proc Natl Acad Sci U S A. 2006 Jul 18;103(29):10883-8. doi: 10.1073/pnas.0604607103. Epub 2006 Jul 10. Proc Natl Acad Sci U S A. 2006. PMID: 16832045 Free PMC article.
-
WNK1 kinase signaling in metastasis and angiogenesis.Cell Signal. 2022 Aug;96:110371. doi: 10.1016/j.cellsig.2022.110371. Epub 2022 May 29. Cell Signal. 2022. PMID: 35649473 Review.
-
Role of an intrinsically disordered conformation in AMPK-mediated phosphorylation of ULK1 and regulation of autophagy.Mol Biosyst. 2012 Jan;8(1):91-6. doi: 10.1039/c1mb05265a. Epub 2011 Aug 18. Mol Biosyst. 2012. PMID: 21853163 Review.
Cited by
-
Genomic Characterization of TP53-Wild-Type Esophageal Carcinoma.Transl Oncol. 2019 Jan;12(1):154-161. doi: 10.1016/j.tranon.2018.09.007. Epub 2018 Oct 11. Transl Oncol. 2019. PMID: 30317074 Free PMC article.
-
The Protective Role of Decorin in Hepatic Metastasis of Colorectal Carcinoma.Biomolecules. 2020 Aug 18;10(8):1199. doi: 10.3390/biom10081199. Biomolecules. 2020. PMID: 32824864 Free PMC article.
-
Identification of Insulin-Mimetic Plant Extracts: From an In Vitro High-Content Screen to Blood Glucose Reduction in Live Animals.Molecules. 2021 Jul 18;26(14):4346. doi: 10.3390/molecules26144346. Molecules. 2021. PMID: 34299620 Free PMC article.
-
Autophagy and beyond: Unraveling the complexity of UNC-51-like kinase 1 (ULK1) from biological functions to therapeutic implications.Acta Pharm Sin B. 2022 Oct;12(10):3743-3782. doi: 10.1016/j.apsb.2022.06.004. Epub 2022 Jun 11. Acta Pharm Sin B. 2022. PMID: 36213540 Free PMC article. Review.
-
UBR5 is a novel regulator of WNK1 stability.Am J Physiol Cell Physiol. 2022 Jun 1;322(6):C1176-C1186. doi: 10.1152/ajpcell.00417.2021. Epub 2022 Apr 20. Am J Physiol Cell Physiol. 2022. PMID: 35442829 Free PMC article.
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
