MLN64 induces mitochondrial dysfunction associated with increased mitochondrial cholesterol content
- PMID: 28282615
- PMCID: PMC5344325
- DOI: 10.1016/j.redox.2017.02.024
MLN64 induces mitochondrial dysfunction associated with increased mitochondrial cholesterol content
Abstract
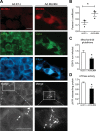
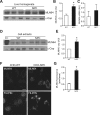
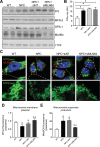
MLN64 is a late endosomal cholesterol-binding membrane protein that has been implicated in cholesterol transport from endosomal membranes to the plasma membrane and/or mitochondria, in toxin-induced resistance, and in mitochondrial dysfunction. Down-regulation of MLN64 in Niemann-Pick C1 deficient cells decreased mitochondrial cholesterol content, suggesting that MLN64 functions independently of NPC1. However, the role of MLN64 in the maintenance of endosomal cholesterol flow and intracellular cholesterol homeostasis remains unclear. We have previously described that hepatic MLN64 overexpression increases liver cholesterol content and induces liver damage. Here, we studied the function of MLN64 in normal and NPC1-deficient cells and we evaluated whether MLN64 overexpressing cells exhibit alterations in mitochondrial function. We used recombinant-adenovirus-mediated MLN64 gene transfer to overexpress MLN64 in mouse liver and hepatic cells; and RNA interference to down-regulate MLN64 in NPC1-deficient cells. In MLN64-overexpressing cells, we found increased mitochondrial cholesterol content and decreased glutathione (GSH) levels and ATPase activity. Furthermore, we found decreased mitochondrial membrane potential and mitochondrial fragmentation and increased mitochondrial superoxide levels in MLN64-overexpressing cells and in NPC1-deficient cells. Consequently, MLN64 expression was increased in NPC1-deficient cells and reduction of its expression restore mitochondrial membrane potential and mitochondrial superoxide levels. Our findings suggest that MLN64 overexpression induces an increase in mitochondrial cholesterol content and consequently a decrease in mitochondrial GSH content leading to mitochondrial dysfunction. In addition, we demonstrate that MLN64 expression is increased in NPC cells and plays a key role in cholesterol transport into the mitochondria.
Copyright © 2017 The Authors. Published by Elsevier B.V. All rights reserved.
Figures



References
-
- Soccio R.E., Breslow J.L. StAR-related lipid transfer (START) proteins: mediators of intracellular lipid metabolism. J. Biol. Chem. 2003;278:22183–22186. - PubMed
-
- Watari H., Arakane F., Moog-Lutz C., Kallen C.B., Tomasetto C., Gerton G.L., Rio M.C., Baker M.E., Strauss J.F., 3rd MLN64 contains a domain with homology to the steroidogenic acute regulatory protein (StAR) that stimulates steroidogenesis. Proc. Natl. Acad. Sci. USA. 1997;94:8462–8467. - PMC - PubMed
-
- Alpy F., Stoeckel M.E., Dierich A., Escola J.M., Wendling C., Chenard M.P., Vanier M.T., Gruenberg J., Tomasetto C., Rio M.C. The steroidogenic acute regulatory protein homolog MLN64, a late endosomal cholesterol-binding protein. J. Biol. Chem. 2001;276:4261–4269. - PubMed
-
- Alpy F., Wendling C., Rio M.C., Tomasetto C. MENTHO, a MLN64 homologue devoid of the START domain. J. Biol. Chem. 2002;277:50780–50787. - PubMed
-
- Alpy F., Tomasetto C. MLN64 and MENTHO, two mediators of endosomal cholesterol transport. Biochem Soc. Trans. 2006;34:343–345. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

