Systemic AAV-Mediated β-Sarcoglycan Delivery Targeting Cardiac and Skeletal Muscle Ameliorates Histological and Functional Deficits in LGMD2E Mice
- PMID: 28284983
- PMCID: PMC5383645
- DOI: 10.1016/j.ymthe.2017.02.013
Systemic AAV-Mediated β-Sarcoglycan Delivery Targeting Cardiac and Skeletal Muscle Ameliorates Histological and Functional Deficits in LGMD2E Mice
Abstract
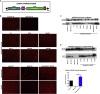
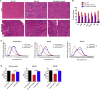
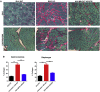
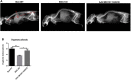
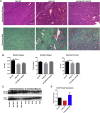
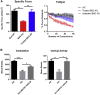
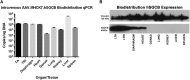
Limb-girdle muscular dystrophy type 2E (LGMD2E), resulting from mutations in β-sarcoglycan (SGCB), is a progressive dystrophy with deteriorating muscle function, respiratory failure, and cardiomyopathy in 50% or more of LGMD2E patients. SGCB knockout mice share many of the phenotypic deficiencies of LGMD2E patients. To investigate systemic SGCB gene transfer to treat skeletal and cardiac muscle deficits, we designed a self-complementary AAVrh74 vector containing a codon-optimized human SGCB transgene driven by a muscle-specific promoter. We delivered scAAV.MHCK7.hSGCB through the tail vein of SGCB-/- mice to provide a rationale for a clinical trial that would lead to clinically meaningful results. This led to 98.1% transgene expression across all muscles that was accompanied by improvements in histopathology. Serum creatine kinase (CK) levels were reduced following treatment by 85.5%. Diaphragm force production increased by 94.4%, kyphoscoliosis of the spine was significantly reduced by 48.1%, overall ambulation increased by 57%, and vertical rearing increased dramatically by 132% following treatment. Importantly, no adverse effects were seen in muscle of wild-type mice injected systemically with scAAV.hSGCB. In this well-defined model of LGMD2E, we have demonstrated the efficacy and safety of systemic scAAV.hSGCB delivery, and these findings have established a path for clinically beneficial AAV-mediated gene therapy for LGMD2E.
Keywords: AAV; LGMD2E; gene therapy; limb-girdle muscular dystrophy; muscular dystrophy; β-sarcoglycan.
Copyright © 2017 The American Society of Gene and Cell Therapy. Published by Elsevier Inc. All rights reserved.
Figures
Similar articles
-
Preclinical Systemic Delivery of Adeno-Associated α-Sarcoglycan Gene Transfer for Limb-Girdle Muscular Dystrophy.Hum Gene Ther. 2021 Apr;32(7-8):390-404. doi: 10.1089/hum.2019.199. Epub 2021 Feb 18. Hum Gene Ther. 2021. PMID: 33349138 Free PMC article. Clinical Trial.
-
β-Sarcoglycan gene transfer decreases fibrosis and restores force in LGMD2E mice.Gene Ther. 2016 Jan;23(1):57-66. doi: 10.1038/gt.2015.80. Epub 2015 Aug 20. Gene Ther. 2016. PMID: 26214262
-
Prevention of cardiomyopathy in delta-sarcoglycan knockout mice after systemic transfer of targeted adeno-associated viral vectors.Cardiovasc Res. 2009 Jun 1;82(3):404-10. doi: 10.1093/cvr/cvp061. Epub 2009 Feb 13. Cardiovasc Res. 2009. PMID: 19218289
-
Sarcoglycanopathies: an update.Neuromuscul Disord. 2021 Oct;31(10):1021-1027. doi: 10.1016/j.nmd.2021.07.014. Epub 2021 Jul 28. Neuromuscul Disord. 2021. PMID: 34404573 Review.
-
Autonomic, locomotor and cardiac abnormalities in a mouse model of muscular dystrophy: targeting the renin-angiotensin system.Exp Physiol. 2014 Apr;99(4):627-31. doi: 10.1113/expphysiol.2013.074336. Epub 2013 Dec 13. Exp Physiol. 2014. PMID: 24334334 Free PMC article. Review.
Cited by
-
Antisense Morpholino-Based In Vitro Correction of a Pseudoexon-Generating Variant in the SGCB Gene.Int J Mol Sci. 2022 Aug 29;23(17):9817. doi: 10.3390/ijms23179817. Int J Mol Sci. 2022. PMID: 36077211 Free PMC article.
-
Current clinical applications of AAV-mediated gene therapy.Mol Ther. 2025 Jun 4;33(6):2479-2516. doi: 10.1016/j.ymthe.2025.04.045. Epub 2025 May 5. Mol Ther. 2025. PMID: 40329530 Review.
-
Advanced Gene-Targeting Therapies for Motor Neuron Diseases and Muscular Dystrophies.Int J Mol Sci. 2022 Apr 27;23(9):4824. doi: 10.3390/ijms23094824. Int J Mol Sci. 2022. PMID: 35563214 Free PMC article. Review.
-
Preclinical Systemic Delivery of Adeno-Associated α-Sarcoglycan Gene Transfer for Limb-Girdle Muscular Dystrophy.Hum Gene Ther. 2021 Apr;32(7-8):390-404. doi: 10.1089/hum.2019.199. Epub 2021 Feb 18. Hum Gene Ther. 2021. PMID: 33349138 Free PMC article. Clinical Trial.
-
Rescue of lysosomal acid lipase deficiency in mice by rAAV8 liver gene transfer.Commun Med (Lond). 2025 Apr 11;5(1):110. doi: 10.1038/s43856-025-00816-8. Commun Med (Lond). 2025. PMID: 40216942 Free PMC article.
References
-
- Moore S.A., Shilling C.J., Westra S., Wall C., Wicklund M.P., Stolle C., Brown C.A., Michele D.E., Piccolo F., Winder T.L. Limb-girdle muscular dystrophy in the United States. J. Neuropathol. Exp. Neurol. 2006;65:995–1003. - PubMed
-
- Bönnemann C.G., Modi R., Noguchi S., Mizuno Y., Yoshida M., Gussoni E., McNally E.M., Duggan D.J., Angelini C., Hoffman E.P. Beta-sarcoglycan (A3b) mutations cause autosomal recessive muscular dystrophy with loss of the sarcoglycan complex. Nat. Genet. 1995;11:266–273. - PubMed
-
- Bönnemann C.G., Passos-Bueno M.R., McNally E.M., Vainzof M., de Sá Moreira E., Marie S.K., Pavanello R.C., Noguchi S., Ozawa E., Zatz M., Kunkel L.M. Genomic screening for beta-sarcoglycan gene mutations: missense mutations may cause severe limb-girdle muscular dystrophy type 2E (LGMD 2E) Hum. Mol. Genet. 1996;5:1953–1961. - PubMed
-
- Araishi K., Sasaoka T., Imamura M., Noguchi S., Hama H., Wakabayashi E., Yoshida M., Hori T., Ozawa E. Loss of the sarcoglycan complex and sarcospan leads to muscular dystrophy in beta-sarcoglycan-deficient mice. Hum. Mol. Genet. 1999;8:1589–1598. - PubMed
-
- Durbeej M., Cohn R.D., Hrstka R.F., Moore S.A., Allamand V., Davidson B.L., Williamson R.A., Campbell K.P. Disruption of the beta-sarcoglycan gene reveals pathogenetic complexity of limb-girdle muscular dystrophy type 2E. Mol. Cell. 2000;5:141–151. - PubMed
Publication types
MeSH terms
Substances
Supplementary concepts
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials

