Lineage Specification from Prostate Progenitor Cells Requires Gata3-Dependent Mitotic Spindle Orientation
- PMID: 28285879
- PMCID: PMC5390093
- DOI: 10.1016/j.stemcr.2017.02.004
Lineage Specification from Prostate Progenitor Cells Requires Gata3-Dependent Mitotic Spindle Orientation
Abstract
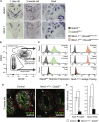
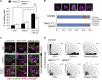
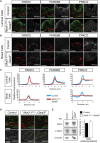
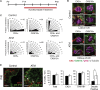
During prostate development, basal and luminal cell lineages are generated through symmetric and asymmetric divisions of bipotent basal cells. However, the extent to which spindle orientation controls division symmetry or cell fate, and the upstream factors regulating this process, are still elusive. We report that GATA3 is expressed in both prostate basal progenitor and luminal cells and that loss of GATA3 leads to a mislocalization of PRKCZ, resulting in mitotic spindle randomization during progenitor cell division. Inherently proliferative intermediate progenitor cells accumulate, leading to an expansion of the luminal compartment. These defects ultimately result in a loss of tissue polarity and defective branching morphogenesis. We further show that disrupting the interaction between PRKCZ and PARD6B is sufficient to recapitulate the spindle and cell lineage phenotypes. Collectively, these results identify a critical role for GATA3 in prostate lineage specification, and further highlight the importance of regulating spindle orientation for hierarchical cell lineage organization.
Keywords: GATA3; atypical protein kinase C; aurothiomalate; cell polarity; epithelial stratification; lineage specification; par complex; prostate development; prostate progenitor cells; spindle orientation.
Copyright © 2017 The Authors. Published by Elsevier Inc. All rights reserved.
Figures




Similar articles
-
Laminin β2 Chain Regulates Retinal Progenitor Cell Mitotic Spindle Orientation via Dystroglycan.J Neurosci. 2018 Jun 27;38(26):5996-6010. doi: 10.1523/JNEUROSCI.0551-18.2018. Epub 2018 May 31. J Neurosci. 2018. PMID: 29853630 Free PMC article.
-
Aurora A kinase regulates mammary epithelial cell fate by determining mitotic spindle orientation in a Notch-dependent manner.Cell Rep. 2013 Jul 11;4(1):110-23. doi: 10.1016/j.celrep.2013.05.044. Epub 2013 Jun 27. Cell Rep. 2013. PMID: 23810554
-
Mammalian aPKC/Par polarity complex mediated regulation of epithelial division orientation and cell fate.Exp Cell Res. 2014 Nov 1;328(2):296-302. doi: 10.1016/j.yexcr.2014.08.008. Epub 2014 Aug 13. Exp Cell Res. 2014. PMID: 25128813 Review.
-
LGN regulates mitotic spindle orientation during epithelial morphogenesis.J Cell Biol. 2010 Apr 19;189(2):275-88. doi: 10.1083/jcb.200910021. Epub 2010 Apr 12. J Cell Biol. 2010. PMID: 20385777 Free PMC article.
-
Cell division orientation and planar cell polarity pathways.Semin Cell Dev Biol. 2009 Oct;20(8):972-7. doi: 10.1016/j.semcdb.2009.03.018. Epub 2009 Apr 7. Semin Cell Dev Biol. 2009. PMID: 19447051 Review.
Cited by
-
Lineage plasticity-mediated therapy resistance in prostate cancer.Asian J Androl. 2019 May-Jun;21(3):241-248. doi: 10.4103/aja.aja_41_18. Asian J Androl. 2019. PMID: 29900883 Free PMC article. Review.
-
Gene signatures predict biochemical recurrence-free survival in primary prostate cancer patients after radical therapy.Cancer Med. 2021 Sep;10(18):6492-6502. doi: 10.1002/cam4.4092. Epub 2021 Aug 28. Cancer Med. 2021. PMID: 34453418 Free PMC article.
-
Claudins are essential for cell shape changes and convergent extension movements during neural tube closure.Dev Biol. 2017 Aug 1;428(1):25-38. doi: 10.1016/j.ydbio.2017.05.013. Epub 2017 May 22. Dev Biol. 2017. PMID: 28545845 Free PMC article.
-
An Integrated Study on the Differential Expression of the FOX Gene Family in Cancer and Their Response to Chemotherapy Drugs.Genes (Basel). 2022 Sep 28;13(10):1754. doi: 10.3390/genes13101754. Genes (Basel). 2022. PMID: 36292640 Free PMC article.
-
Regulation of stem/progenitor cell maintenance by BMP5 in prostate homeostasis and cancer initiation.Elife. 2020 Sep 7;9:e54542. doi: 10.7554/eLife.54542. Elife. 2020. PMID: 32894216 Free PMC article.
References
-
- Asselin-Labat M.L., Sutherland K.D., Barker H., Thomas R., Shackleton M., Forrest N.C., Hartley L., Robb L., Grosveld F.G., van der Wees J. Gata-3 is an essential regulator of mammary-gland morphogenesis and luminal-cell differentiation. Nat. Cell Biol. 2007;9:201–209. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases

