Adipose tissue fibrosis in human cancer cachexia: the role of TGFβ pathway
- PMID: 28288584
- PMCID: PMC5348844
- DOI: 10.1186/s12885-017-3178-8
Adipose tissue fibrosis in human cancer cachexia: the role of TGFβ pathway
Abstract
Background: Cancer cachexia is a multifactorial syndrome that dramatically decreases survival. Loss of white adipose tissue (WAT) is one of the key characteristics of cachexia. WAT wasting is paralleled by microarchitectural remodeling in cachectic cancer patients. Fibrosis results from uncontrolled ECM synthesis, a process in which, transforming growth factor-beta (TGFβ) plays a pivotal role. So far, the mechanisms involved in adipose tissue (AT) re-arrangement, and the role of TGFβ in inducing AT remodeling in weight-losing cancer patients are poorly understood. This study examined the modulation of ECM components mediated by TGFβ pathway in fibrotic AT obtained from cachectic gastrointestinal cancer patients.
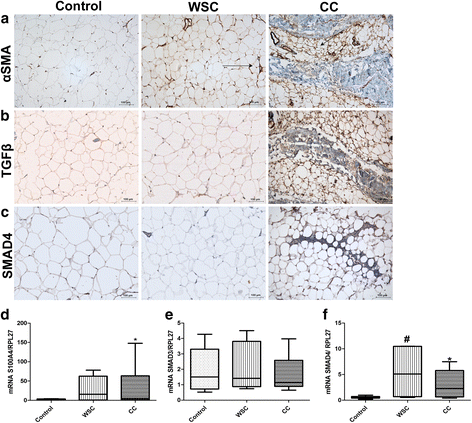
Methods: After signing the informed consent form, patients were enrolled into the following groups: cancer cachexia (CC, n = 21), weight-stable cancer (WSC, n = 17), and control (n = 21). The total amount of collagen and elastic fibers in the subcutaneous AT was assessed by histological analysis and by immunohistochemistry. TGFβ isoforms expression was analyzed by Multiplex assay and by immunohistochemistry. Alpha-smooth muscle actin (αSMA), fibroblast-specific protein (FSP1), Smad3 and 4 were quantified by qPCR and/or by immunohistochemistry. Interleukin (IL) 2, IL5, IL8, IL13 and IL17 content, cytokines known to be associated with fibrosis, was measured by Multiplex assay.
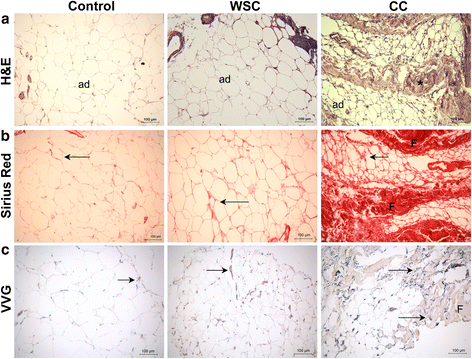
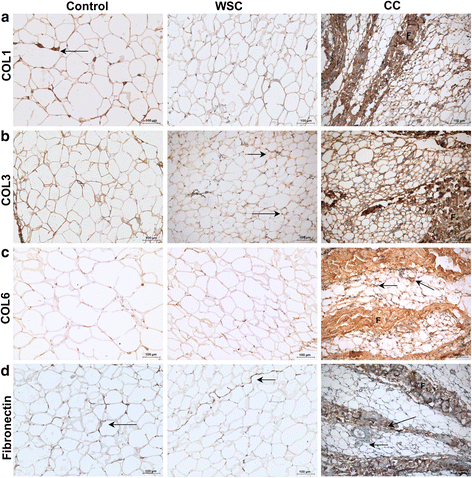
Results: There was an accumulation of collagen and elastic fibers in the AT of CC, as compared with WSC and controls. Collagens type I, III, VI, and fibronectin expression was enhanced in the tissue of CC, compared with both WSC and control. The pronounced expression of αSMA in the surrounding of adipocytes, and the increased mRNA content for FSP1 (20-fold) indicate the presence of activated myofibroblasts; particularly in CC. TGFβ1 and TGFβ3 levels were up-regulated by cachexia in AT, as well in the isolated adipocytes. Smad3 and Smad4 labeling was found to be more evident in the fibrotic areas of CC adipose tissue.
Conclusions: Cancer cachexia promotes the development of AT fibrosis, in association with altered TGFβ signaling, compromising AT organization and function.
Keywords: Adipose tissue; Cancer cachexia; Extracellular matrix; Fibrosis; TGFβ.
Figures

References
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous

