Functional Organization of Vestibulo-Ocular Responses in Abducens Motoneurons
- PMID: 28292832
- PMCID: PMC6596586
- DOI: 10.1523/JNEUROSCI.2626-16.2017
Functional Organization of Vestibulo-Ocular Responses in Abducens Motoneurons
Abstract
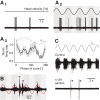
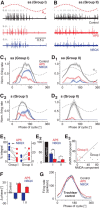
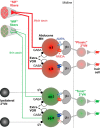
Vestibulo-ocular reflexes (VORs) are the dominating contributors to gaze stabilization in all vertebrates. During horizontal head movements, abducens motoneurons form the final element of the reflex arc that integrates visuovestibular inputs into temporally precise motor commands for the lateral rectus eye muscle. Here, we studied a possible differentiation of abducens motoneurons into subtypes by evaluating their morphology, discharge properties, and synaptic pharmacology in semi-intact in vitro preparations of larval Xenopus laevis Extracellular nerve recordings during sinusoidal head motion revealed a continuum of resting rates and activation thresholds during vestibular stimulation. Differences in the sensitivity to changing stimulus frequencies and velocities allowed subdividing abducens motoneurons into two subgroups, one encoding the frequency and velocity of head motion (Group I), and the other precisely encoding angular velocity independent of stimulus frequency (Group II). Computational modeling indicated that Group II motoneurons are the major contributor to actual eye movements over the tested stimulus range. The segregation into two functional subgroups coincides with a differential activation of glutamate receptor subtypes. Vestibular excitatory inputs in Group I motoneurons are mediated predominantly by NMDA receptors and to a lesser extent by AMPA receptors, whereas an AMPA receptor-mediated excitation prevails in Group II motoneurons. Furthermore, glycinergic ipsilateral vestibular inhibitory inputs are activated during the horizontal VOR, whereas the tonic GABAergic inhibition is presumably of extravestibular origin. These findings support the presence of physiologically and pharmacologically distinct functional subgroups of extraocular motoneurons that act in concert to mediate the large dynamic range of extraocular motor commands during gaze stabilization.SIGNIFICANCE STATEMENT Outward-directed gaze-stabilizing eye movements are commanded by abducens motoneurons that combine different sensory inputs including signals from the vestibular system about ongoing head movements (vestibulo-ocular reflex). Using an amphibian model, this study investigates whether different types of abducens motoneurons exist that become active during different types of eye movements. The outcome of this study demonstrates the presence of specific motoneuronal populations with pharmacological profiles that match their response dynamics. The evolutionary conservation of the vestibulo-ocular circuitry makes it likely that a similar motoneuronal organization is also implemented in other vertebrates. Accordingly, the physiological and pharmacological understanding of specific motoneuronal contributions to eye movements might help in designing drug therapies for human eye movement dysfunctions such as abducens nerve palsy.
Keywords: GABA; extraocular motoneurons; glutamate; glycine; semicircular canal; vestibulo-ocular reflex.
Copyright © 2017 the authors 0270-6474/17/374032-14$15.00/0.
Figures






References
-
- Broussard DM, DeCharms RC, Lisberger SG (1995) Inputs from the ipsilateral and contralateral vestibular apparatus to behaviorally characterized abducens neurons in rhesus monkeys. J Neurophysiol 74:2445–2459. - PubMed
-
- Büttner-Ennever JA, Horn AK (2002) Oculomotor system: a dual innervation of the eye muscles from the abducens, trochlear, and oculomotor nuclei. Mov Disord 17:S2–S3. - PubMed
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources
Other Literature Sources
